New and Improved Ways to Create Stem Cells
Stem cells start out as a blank slate but eventually, they give rise to all the different kinds of specialized cells that the different structures and tissues of organisms need. Stem cells held tremendous therapeutic potential but were stymied because of ethical issues in harvesting them. Then, the 2012 Nobel in Medicine went to a technique that could create stem cells from specialized cells. Genes can be added to mature skin cells, and that changes them into a generic stem cell in a process called reprogramming. Now, researchers have improved on that method, and found a totally new way to reprogram cells.
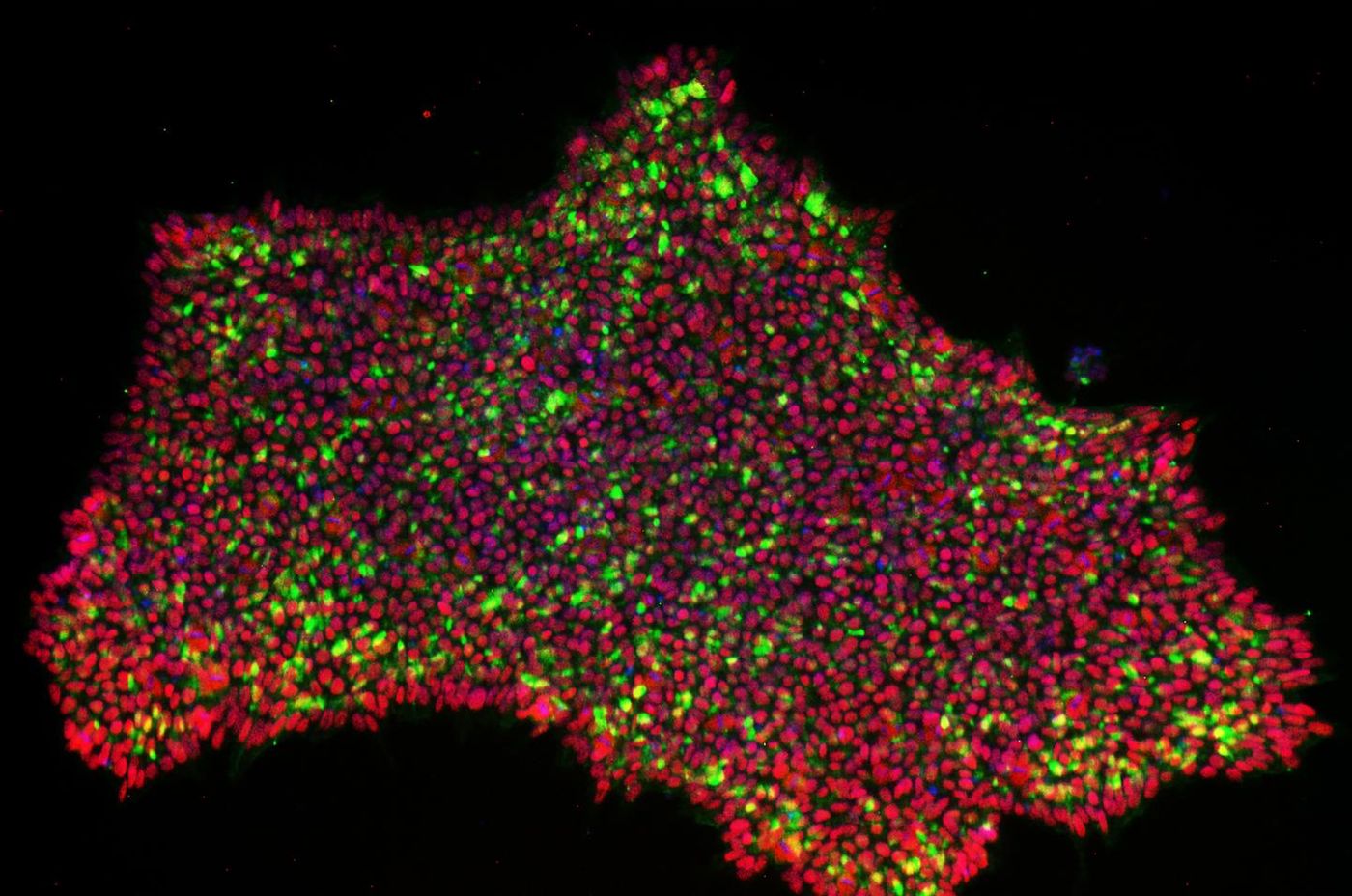
Reported in Nature Communications, one technique utilizes the Yamanaka factors identified in the Nobel-winning work. But instead of adding the necessary genes to cells to initiate the reprogramming, this new technique uses a modification of the genetic editing tool CRISPR to use the cell's own genome to express those critical genes. The work was led by Professor Timo Otonkoski at the University of Helsinki and Professor Juha Kere of Karolinska Institutet and King's College London.
"CRISPR/Cas9 can be used to activate genes. This is an attractive possibility for cellular reprogramming because multiple genes can be targeted at the same time. Reprogramming based on activation of endogenous genes rather than overexpression of transgenes is also theoretically a more physiological way of controlling cell fate and may result in more normal cells. In this study, we show that it is possible to engineer a CRISPR activator system that allows robust reprogramming of iPSC," explained Professor Otonkoski.
The scientists found they also had to activate a genetic element that normally helps regulate the development of the human embryo. "Using this technology, pluripotent stem cells were obtained that resembled very closely typical early embryonal cells," said Professor Kere.
This research may have other applications in cell reprogramming. "The technology may find practical use in biobanking and many other tissue technology applications," said first author Jere Weltner, graduate student. "In addition, the study opens up new insights into the mechanisms controlling early embryonic gene activation."
The researcher that led the Nobel winning work, Shinya Yamanaka, is featured in the video below.
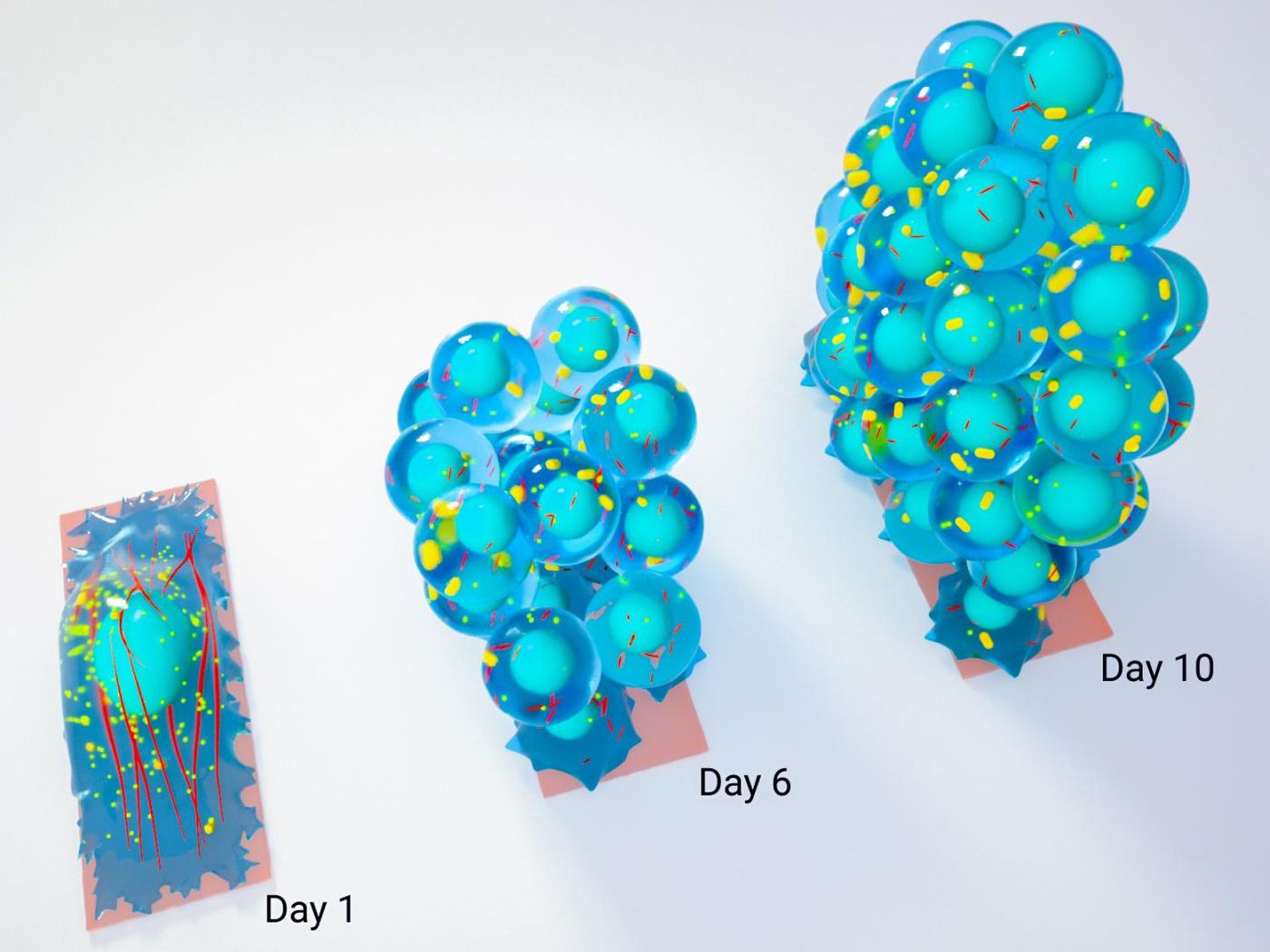
Another new tool to create stem cells has been reported in the Proceedings of the National Academy of Sciences (PNAS). This technique uses a physical method to transform mature cells into stem cells. When cells are constrained into a geometric form, it makes them take on the properties of a stem cell. The researchers believe that this work will have a big impact.
"Our breakthrough findings will usher in a new generation of stem cell technologies for tissue engineering and regenerative medicine that may overcome the negative effects of genomic manipulation," said study leader Professor G.V. Shivashankar. He holds appointments at the Mechanobiology Institute (MBI) of the National University of Singapore (NUS) and the FIRC Institute of Molecular Oncology (IFOM) in Italy.
While it’s known that cells can be transformed into pluripotent stem cells with genetic tools, using the Yamanaka method, when those transformed cells are used therapeutically, they have a tendency to start growing tumors.
The scientists tested their method and found that the cells they grew in rectangular boxes had taken on the genetic characteristics of a ten-day-old embryonic stem cell. When the cells are confined in this way for a longer period, they are transformed into pluripotent stem cells.
"While it is well established that confining stem cells to defined geometric patterns and substrate properties can direct their differentiation into specialized cells, this study shows for the first time that mechanical cues can reset the genomic program of mature cells and return them to a pluripotent state," Professor Shivashankar explained.
"The use of geometric constraints to reprogram mature cells may better reflect the process occurring naturally within the body," he added. "More importantly, our findings allow researchers to generate stem cells from mature cells with high efficiency and without genetically modifying them."
Sources: Phys.org, University of Helsinki, National University of Singapore, Nature Communications, PNAS