Preventing the Wrong Cells From Forming in Organoids
Researchers have long relied on models to study biology in the lab. Cells are a commonly used model; they are often grown in a layer in a dish or liquid suspension in a flask. But that is not how cells typically grow in a biological organism, where they are formed into three-dimensional structures and systems. In an effort to develop a more accurate model of human biology, researchers have created organoids -small, three-dimensional constructs of cells that represent a simplified version of a full-size organ. Organoids have already helped researchers learn more about health and disease. But are they what they seem?
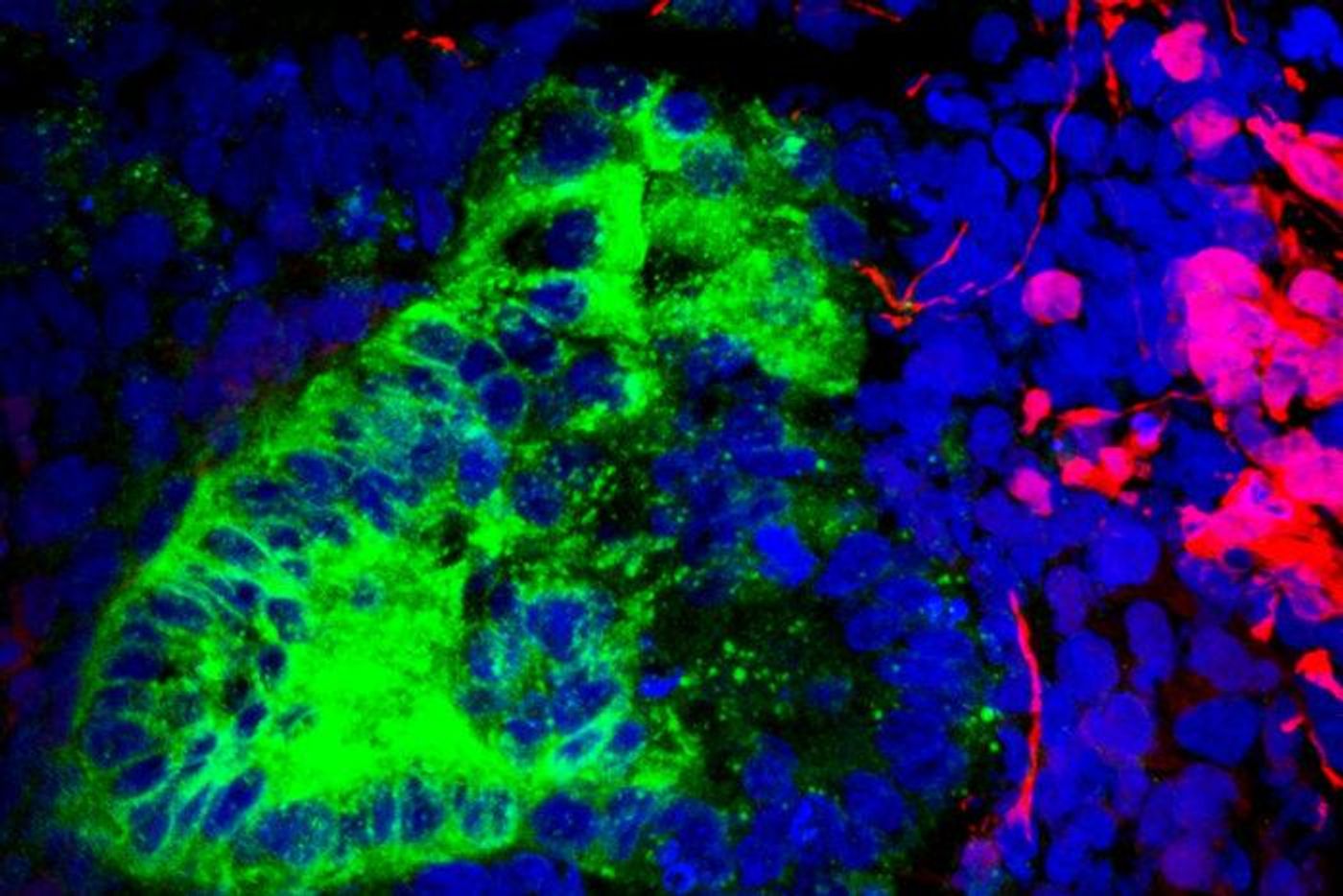
Organoids are generated from stem cells and are often used in the development of treatments, especially for kidney disease. Scientists at Washington University School of Medicine in St. Louis (WUSTL) wanted to test kidney organoids that were created with two different protocols to verify that they were representative of the human organ. They found rogue cells lurking in kidney organoids, however. The researchers identified them as brain and muscle cells, showing that the methods used to turn stem cells into kidney cells are imperfect.
The study, which was reported in Cell Stem Cell, showed that around ten to twenty percent of organoid cells were not the proper cell type for the kidney organoid. It’s not time to abandon organoids though. The scientists found a straightforward way to stop those aberrant cells from growing, a process that may also apply to other types of organoids.
"There's a lot of enthusiasm for growing organoids as models for diseases that affect people," said Benjamin D. Humphreys, MD, Ph.D., senior study author and director of the Division of Nephrology at WUSTL. "But scientists haven't fully appreciated that some of the cells that make up those organoids may not mimic what we would find in people. The good news is that with a simple intervention, we could block most of the rogue cells from growing. This should really accelerate our progress in making organoids better models for human kidney disease and drug discovery, and the same technique could be applied to targeting rogue cells in other organoids."
Related: Lung Organoids are Similar to the Full-size Organ
There is a dire need for better kidney disease treatment in the US; an estimated 500,000 people have end-stage kidney disease and are currently receiving dialysis.
"Developing kidney organoids is driven by the reality that we have so many patients with failing kidneys and no effective drugs to offer them," noted Humphreys.
The two protocols used to generate kidney organoids both start with stem cells; one uses induced pluripotent stem cells, which are made from mature adult cells, while the other uses embryonic stem cells. A molecular cocktail triggers the cells to grow into kidney cells. After allowing the cells to become specialized for four weeks, the investigators did an in-depth analysis to see what had grown.
In a humongous single-cell RNA sequencing project, they assessed gene expression in 83,130 cells harvested from 65 kidney organoids.
"This generates massive amounts of data, and there's no way our brains can make sense of it all," Humphreys admitted. "But computers can easily compare gene activity across 83,000 cells and, using artificial intelligence, group cell types together based on their gene expression. So rather than looking for cells that we think we thought we'd find in the organoid, it helped us find cells even if we'd never imagined they'd be there."
The team found that ten to twenty percent of the organoid cells did not develop as kidney cells, becoming brain or muscle tissue instead, regardless of which method was used. The researchers were able to find the point at which the cells missed their cue, however. By blocking unwanted cells from forming, the team reduced the level of brain cells in the kidney organoids by 90 percent. The work also shows how this technique can be applied to other organoids.
"Progress to develop better treatments for kidney disease is slow because we lack good models," Humphreys said. "We rely on mice and rats, and they are not little humans. There are many examples of drugs that have done magically well at slowing or curing kidney disease in rodents but failed in clinical trials. So, the notion of channeling human stem cells to organize into a kidney-like structure is tremendously exciting because many of us feel that this potentially eliminates that 'lost in translation' aspect of going from a mouse to a human.”
Learn more about how kidney organoids are helping researchers understand disease from the video.
Sources: Science Daily via WUSTL, Cell Stem Cell