"Silent" RNA Increases Progression of COPD
Chronic Obstructive Pulmonary Disease or COPD is a prevalent lung disease among the elderly population. It is characterized by “emphysema, chronic bronchitis, asthma,” and many others. It is most commonly found in the aging population who have smoked or currently smoke. It is unknown what causes chronic inflammation in the lungs of COPD patients, and while there are treatment options, there is not a cure for this disease. When treatment is given to repair the damaged lungs, the lungs repair abnormally and don't allow optimal function.
To address this critical health concern, Wenchao Gu and their team at the Second Military Medical University in Shanghai, China studied “long non-coding RNAs (lncRNAs).” They aimed to find a relationship with gene expression controlled by these "silent" lncRNAs and the inflammation response within the lungs.
lncRNAs are a class of regulatory RNA. They do provide the genetic code to make proteins themselves but affect gene expression. As more research is done to study their function, they could be used as potential biomarkers for disease and drug targets. The lncRNA of choice is known as TUG1. TUG1 is associated with the progression of many human diseases. TUG1, along with other lncRNA’s over abnormally expressed during the disease progression. Gu reveals “that TUG1 expression is markedly higher in COPD-affected lung tissues compared to that in non-COPD lung tissues.”
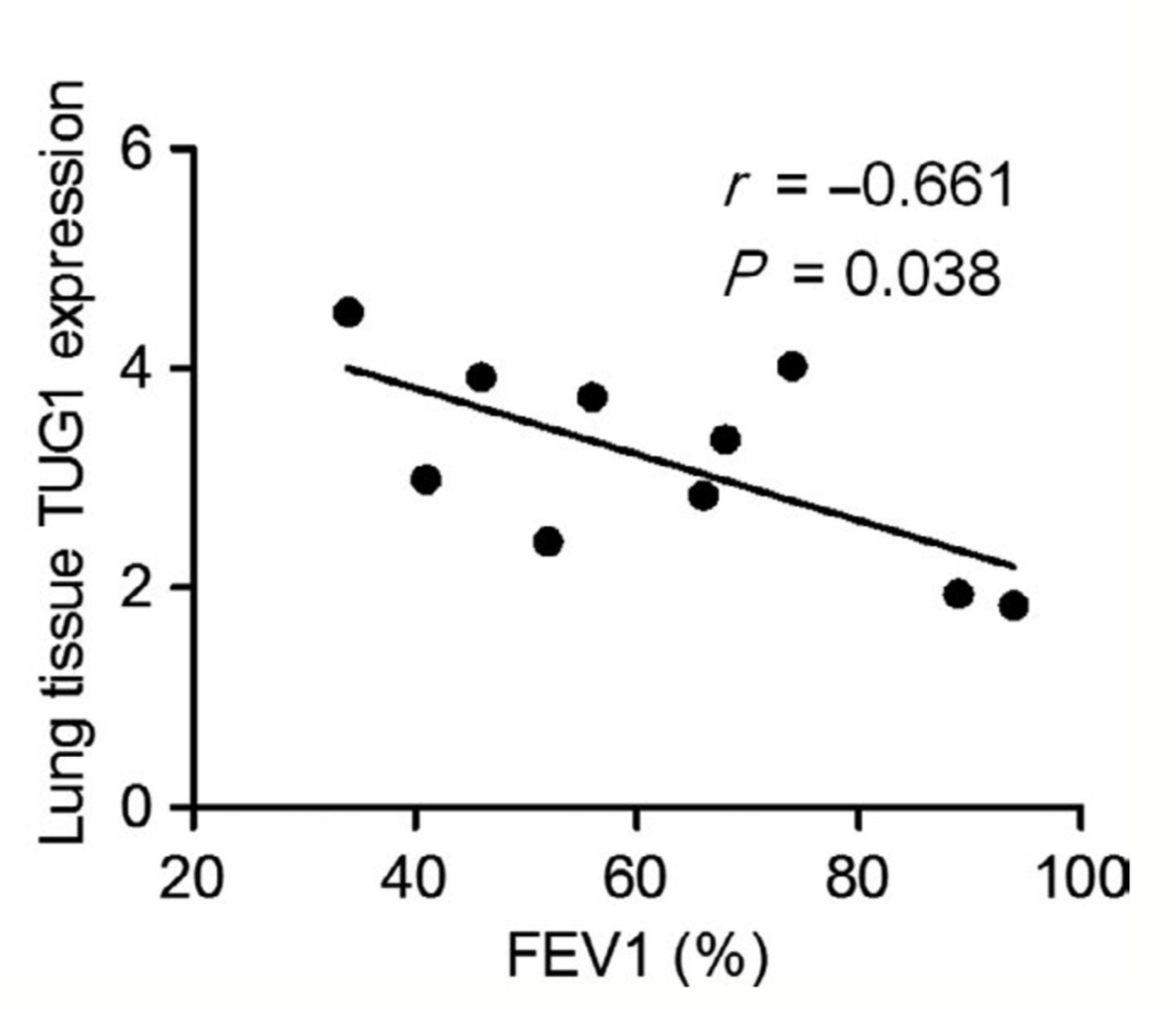
An important marker of the progressions and stage of COPD within a patient is the FEV1 value. This value represents the amount of air that a person can force out of their lungs in a 1 second period. Lower than average FEV1 values indicate the progression and severity of the COPD. A higher the concentration of TUG1 correlated with a lower FEV1 value, as shown in the figure from the study below:
TUG1 was also shown to increase in patients with COPD compared to non-smokers. These results suggest that the TUG1 expression correlates with lower FEV1 values which are indicative of COPD. When the researchers restricted the action of TUG1, they found that inflammation within the airway decreased.
Gu and their team reveal an essential molecular mechanism of TUG1. TUG1 was shown to bind to other RNA that directly effected the inflammatory response. When the function of TUG1 was inhibited, the endogenous RNA reversed its role and decreased the inflammatory response.
This study is just one critical step in discovering the cause, progression, and molecular mechanisms of COPD. Eventually, this study and others like it will allow for “lncRNA-directed diagnostics and therapeutics of COPD.”
To learn more lncRNA, watch the video below:
To learn more about COPD, watch the video below:
Sources: