Antibiotic-Resistance Genes Can Move From Humans to Animals
Researchers at Clemson University have been investigating how antibiotic resistance can move between species, including from humans into animals. Infectious diseases that can jump species are called zoonotic diseases. Tens of thousands of times every year, people get sick after contracting an illness from direct contact with an animal or through food or water; examples include Salmonella, anthrax and E. coli. But illnesses are not the only things that can be transmitted between species when these bacteria move. Genes that confer antibiotic resistance can move as well.
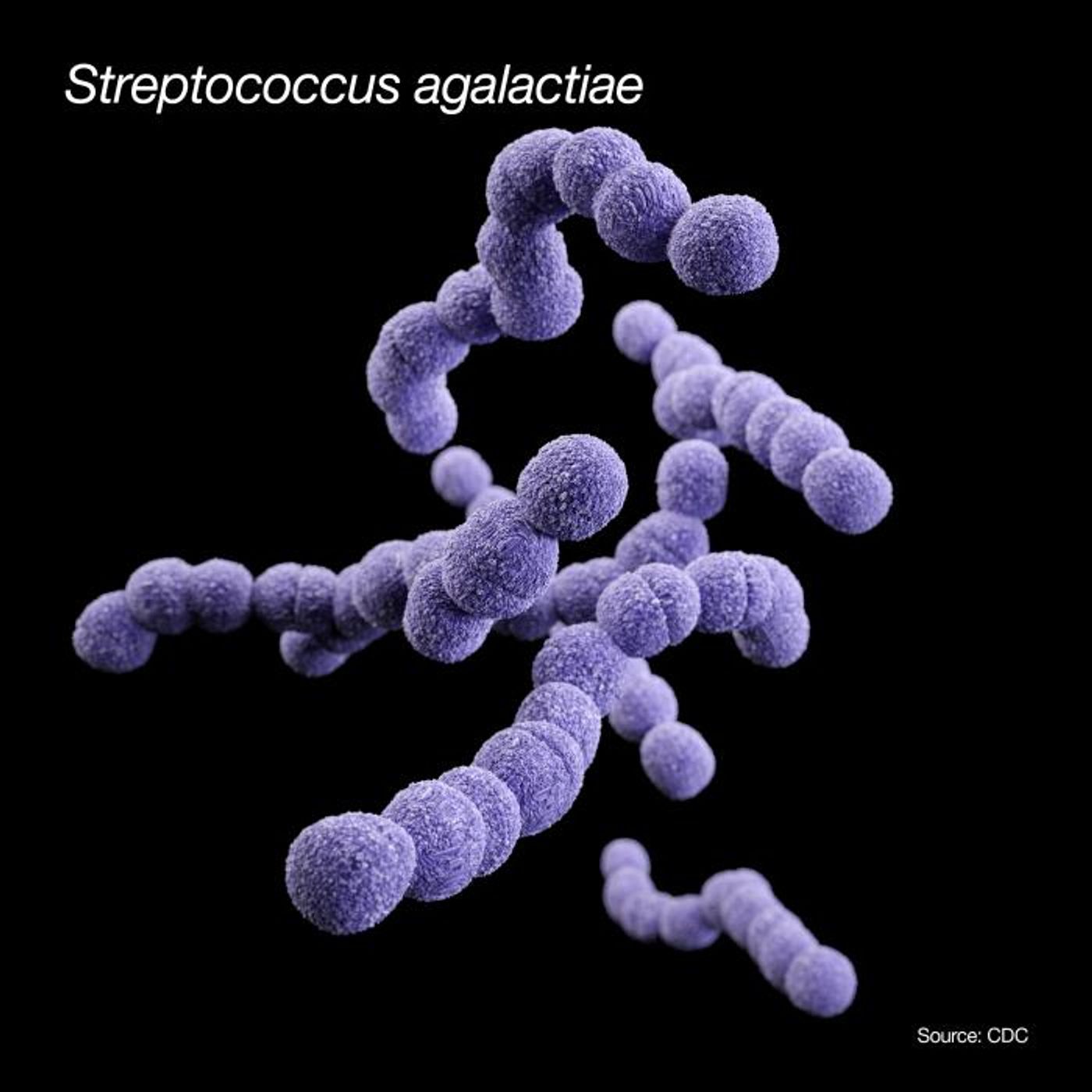
"I found actual cases of transmission of these antibiotic resistance genes from humans into livestock, companion animals and wildlife," said Vincent Richards, who suggested that the genes are using wastewater as a conduit, or that the genes move during the handling of animals. In new work reported in Molecular Biology and Evolution, his team studied how the pathogenic bacterium Streptococcus agalactiae (or group B Strep) moves among different species.
"One of the things that makes the bacteria so interesting is its wide host range," noted Richards, an assistant professor in the department of biological sciences at Clemson. "It doesn't just infect humans and cows, it infects a whole range of terrestrial and aquatic mammals, reptiles and amphibians, and fish. It has quite the taxonomic range for a bacteria."
In this work, the researchers looked at 901 different genomic sequences of the bacteria harvested from nine different species that the bacterium infects - humans, dolphins, dogs, fish, frogs, gray seals, cows, goats,and a camel - to gain insight into the transmission process. In humans, group B Strep can cause serious illnesses including pneumonia, meningitis, and sepsis in newborns. The bacterium also causes an inflammatory disease in cows that disrupts milk production, called bovine mastitis.
The genes that were revealed in their analysis were grouped into core categories - genes shared by all genomes, and dispensable genes - found only in some of the species’ genomes. Taken together, they represent the pan-genome, encompassing all genes found in all strains of the bacteria. Richards was surprised to find that about 90 percent of the genes in the pan-genome fell into the dispensable group, leaving only the remaining ten percent as core genes.
The researchers also found that genes conferring resistance to the antibiotic tetracycline, which likely originates in humans due to the unnecessary or overuse of antibiotics, were moving into animals.
The work illustrates how flexible the bacterial genome can be; the pan-genome is expansive, and can continue to diversify. This plasticity enables bacteria to rapidly adapt to changes or to new environments, generating many diverse populations. This variability can also show how genes that arose in one host or environment can move to another one.
"You have genes that are adapted to a particular population now being transferred into another population," Richards said. "A particular gene for antibiotic resistance that was selected in human populations is now being transmitted into animal populations."
Richards wants to continue this work on related strep bacteria that can be found in the human mouth, and specifically, how certain genes play a role in tooth decay in children.
In other work, researchers have found that in general, antibiotic-resistance is rising in livestock. Learn more from the video.
Sources: AAAS/Eurekalert! Via Clemson University, Molecular Biology and Evolution