Scientists Find a Non-Invasive Way to Detect Prions
Our cells use coding regions of genes to generate proteins, which are necessary for biological structure and function. Proteins are made of strings of amino acids, which have to be folded into the correct shape so they do their job properly. Misfolded proteins, also called prions, can cause a host of problems, including neurodegenerative disorders. For many years, however, there was no good way to test for these prions. New work reported in PLOS One will change that.
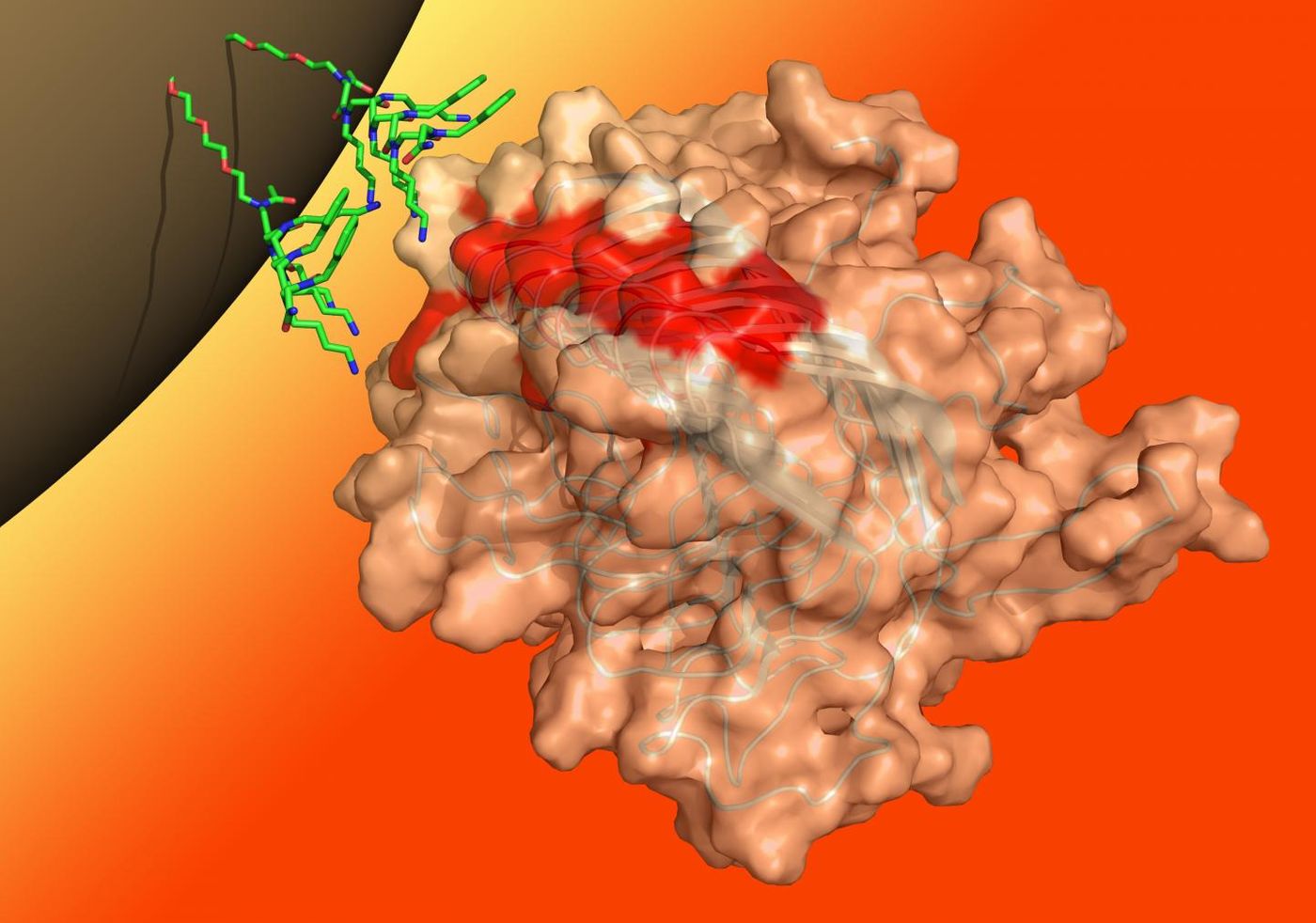
The technique is the product of twenty years of work, and uses synthetic molecules to isolate prions from bodily fluids. The researchers used samples from animals infected with prions to show that it works, and is now the only non-invasive way to detect prions in live subjects. The assay utilizes artificial compounds called peptides that cover tiny magnets. When placed in a liquid sample, the peptoid-beads link up with misfolded proteins and ignore properly folded ones.
"It's like Velcro," said the study co-author Michael Connolly, senior scientific engineer of the Molecular Foundry's biological nanostructures group. "The aggregated misfolded protein has multiple hooks - multiple binding sites - that will attach to the bead, which is like the complementing sheet. But the natural, correctly folded protein only has a single hook, so its binding affinity is much less." Once the beads are bound to prions, they can be removed from the liquid sample magnetically, and tested with a misfolded protein assay (MPA).
"Our peptoid beads have the ability to detect the misfolded proteins that act as infectious agents, so it could have a significant impact in the realm of prion diseases, but we have also shown that it can seek out the large aggregated proteins that are the disease agents in Alzheimer's and Parkinson's diseases, among others" said Ronald Zuckermann, a senior scientist at Lawrence Berkeley National Laboratory (Berkeley Lab)'s Molecular Foundry."Prion diseases are rare, but there are many misfolded protein-based diseases, which affect millions of people, that are also very poorly understood. And like prion diseases, we need a way to diagnose these slow-onset conditions in the years before symptoms arise."
The test may have a wide range of applications. "On top of potentially detecting asymptomatic disease carriers, the peptoid-bead MPA could be optimized to screen blood and blood products in a cheap and fast manner, a capability that will be very important for avoiding accidental transmission in case of a new prion disease outbreak," said the first author of the study Simone Hornemann, a senior researcher in the lab of neuropathologist and prion expert Adriano Aguzzi at the University Hospital Zurich's Institute of Neuropathology. "This assay could also be modified to test deer and elk for chronic wasting disease (CWD), a prion disease that is considered as a global epidemic in these animals."
Prion diseases can impact many different animals, and these disorders, called transmissible spongiform encephalopathies, or TSEs occur when prions take on a certain shape. Through an as-yet-unknown mechanism, they can begin to cause normal proteins to take on a similar shape and clump together. These aggregates begin to build up in the nervous system and cause cells to die. TSEs might arise from exposure to misfiled proteins from another individual, or from an inherited genetic mutation.
Research on prion diseases began many years ago, and the misfolded proteins were identified in 1985. In the late 1980s, mad cow disease, also called bovine spongiform encephalopathy gave the studies new urgency. The prions are present only in low concentrations, making them very tough to detect.
"Back in the '90s, we were at a company, now part of Novartis, that was interested in detecting these types of diseases in blood, and at the time this seemed pretty farfetched," said Zuckermann. "The diagnostic task sounded straightforward because this is an aggregate of the same protein wanting to link to itself, so if you just stick one of its peptides on a bead or an antibody then it should bind automatically."
It took time, but the research team developed a way to mass-produce any kind of peptoids. "We recognized from the outset that their utility went a lot further than just prions," continued Zuckermann. "So Michael and I set up a resource here at Berkeley Lab where we can design and produce peptoids for any field, whether it's biomedical, energy, or nanomaterials."
Sources: AAAS/Eurekalert! via DOE/Lawrence Berkeley National Laboratory, PLOS One