A Shield Surrounds the Flexible SARS-CoV-2 Spike Protein
Researchers know that the viruses including SARS-CoV-2 have a spike protein that allows them to bind to receptors on host cells. In the case of SARS-CoV-2, ACE2 receptors are among those that can bind to the SARS-CoV-2 spike protein, which allows the virus to bind to the host cell membrane and release its contents inside, infecting the host cell.
Scientists have wanted to learn as much as they can about the Sars-CoV-2 spike protein, because it's where an infection starts, and its characteristics could have critical implications for the development of vaccines and therapeutics; this protein triggers an immune response in people.
Researchers have now used advanced techniques including molecular dynamics simulations and cryo-electron tomography, a tool for generating high-resolution three-dimensional views of macromolecules to assess the molecular structure of the spike protein at nearly an atomic scale. The findings have been reported in Science.
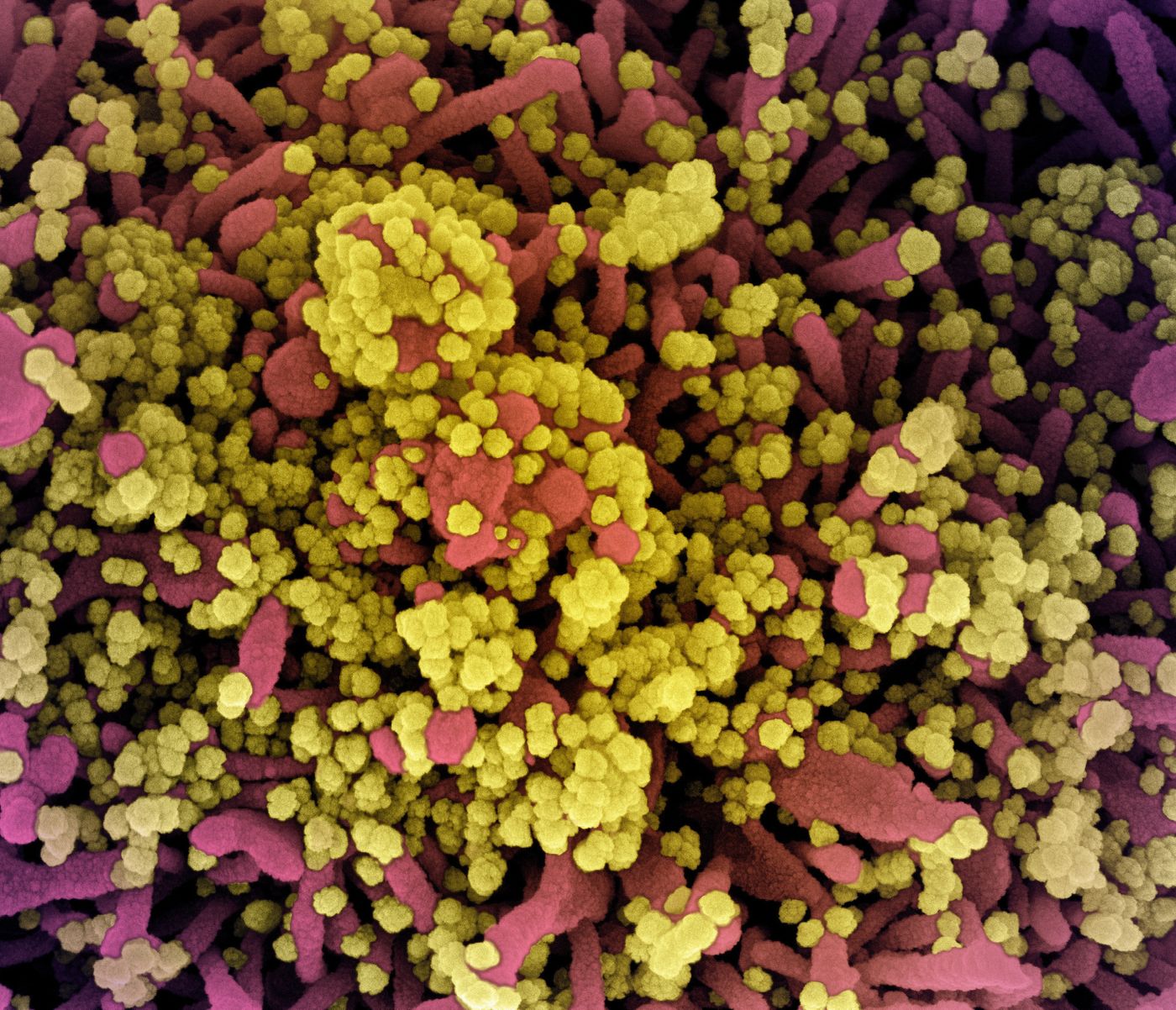
The investigators found that the SARS-CoV-2 spike protein has a spherical portion, which contains regions that bind and fuse to the host cell receptor. This is linked to a flexible stalk.
"The upper spherical part of the spike has a structure that is well reproduced by recombinant proteins used for vaccine development," explained Martin Beck, European Molecular Biology Laboratory (EMBL) group leader and a director of the Max Planck Institute (MPI) of Biophysics. "However, our findings about the stalk, which fixes the globular part of the spike protein to the virus surface, were new."
"The stalk was expected to be quite rigid," added Gerhard Hummer, of the MPI of Biophysics and the Institute of Biophysics at Goethe University Frankfurt. "But in our computer models and in the actual images, we discovered that the stalks are extremely flexible."
In this work, 266 cryo-tomograms of about 1,000 different viruses, each carrying about 40 spikes apiece, were created. After dynamics simulations and subtomogram averaging - an image processing tool that combines the tomograms, the new structural information about the spike protein was revealed. The flexibility of the stalk lies with three 'joints' that act as its ankle, knee, and hip.
"Like a balloon on a string, the spikes appear to move on the surface of the virus and thus are able to search for the receptor for docking to the target cell," said Jacomine Krijnse Locker, group leader at the Paul-Ehrlich-Institut.
Antibodies targeting the spike protein can prevent infection, neutralizing the virus, if they attach to the spike protein and prevent it from binding to a host cell. This work has shown that the entire spike protein, including the stalk, carries chains of sugar molecules called glycans. This gives the spike a kind of protective shield that helps prevent such neutralizing antibodies from binding to it.
Sources: AAAS/Eurekalert! via European Molecular Biology Laboratory, Science