Modeling Embryonic Development in the Laboratory
Embryonic development is a critical process for all organisms including humans. Many different diseases can arise when there are problems during development, but it can be extremely challenging to study the underlying physiological processes. There are some transparent animal models for that purpose, like fish or frogs, and early mammalian embryos can be isolated and grown in a dish, but mammals have to eventually implant in the uterus to continue growing, which obscures the activity.
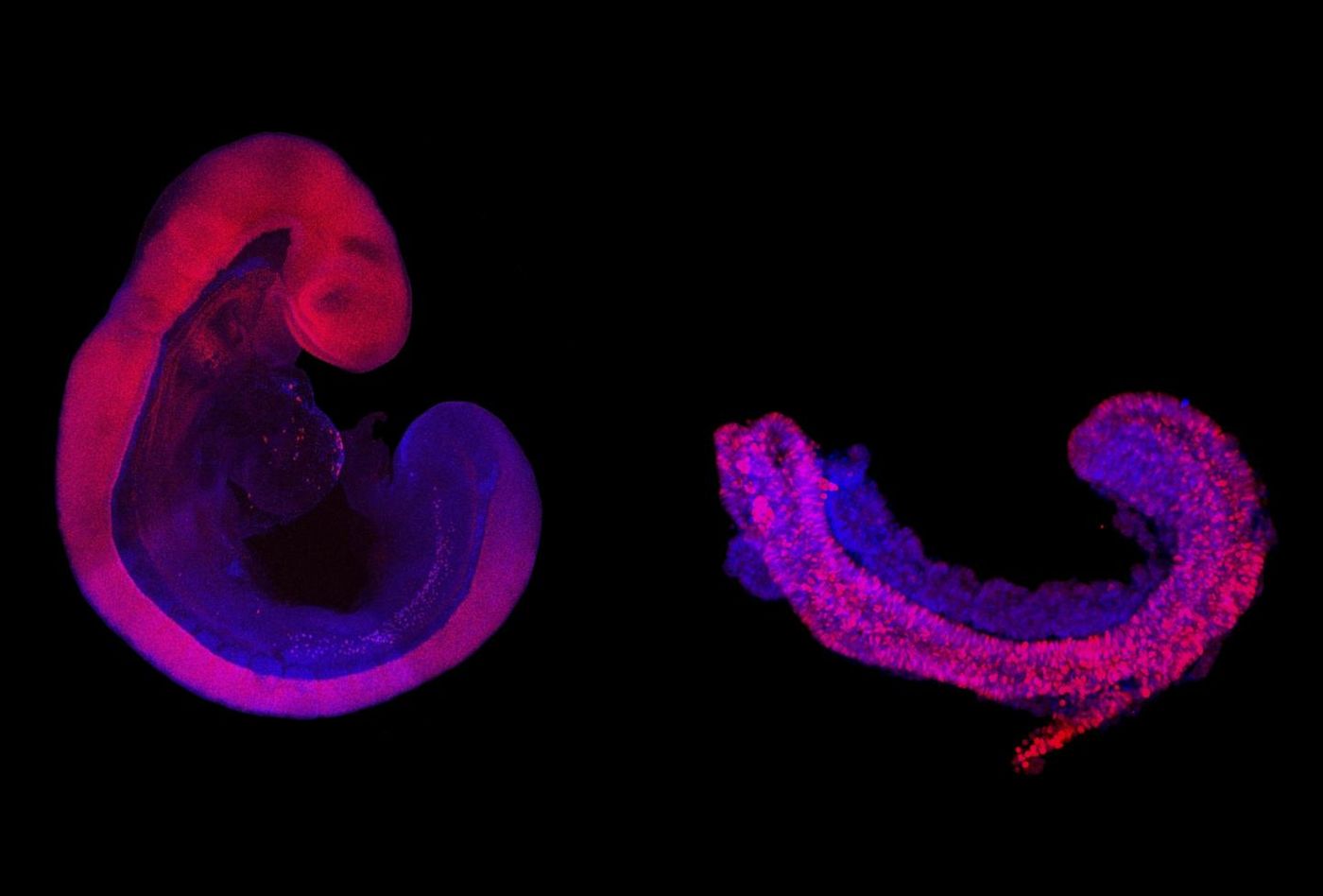
Now researchers have successfully coaxed cells within a special gel to grow into the structures that give rise to bone, cartilage, muscle, and neuronal tissues. These three-dimensional structures are about a millimeter in size and contain a model of a neural tube that is meant to mimic the one that would generate a spinal cord. Some of these 3D models or 'trunk-like structures' produce somites, the precursors of skeleton and muscle, as well as internal organ precursors. The process lasts for about five days before the structures no longer parallel normal development.
"This model of embryonic development starts a new era," said joint study supervisor Bernhard G. Herrmann, Director of the Max Planck Institute for Molecular Genetics (MPIMG) and head of the Institute of Medical Genetics at Charité - Universitätsmedizin. "This allows us to observe embryogenesis of the mouse directly, continuously, and with large parallel numbers of samples, which would not be possible in the animal."
This work, which was reported in Science, will advance the study of development without the need for animal embryos, and may one day help scientists create strategies for repairing errors that arise.
"We can obtain more detailed results more quickly, and without the need for animal research," said joint study supervisor Alexander Meissner, Director at the MPIMG. "Of the more complex processes such as morphogenesis, we usually only get snapshots, but this changes with our model."
By using a gel, the researchers were able to provide the embryonic models with an environment that mimics the extracellular matrix. This complex mix of molecules has been the 'special sauce' for this new method, providing a framework for growth and preventing important molecules from diffusing away into the culture medium.
"The gel provides support to the cultured cells and orients them in space; they can distinguish inside from outside, for example," said co-lead study author Jesse Veenvliet. "The cells are able to establish better communication, which leads to better self-organization."
In this study, the researchers analyzed the structures at the single-cell level after five days of growth.
"Even though not all cell types are present in the trunk-like structures, they are strikingly similar to an embryo of the same age," said co-lead study author Adriano Bolondi.
Bolondi and colleagues compared the structures' gene expression patterns with those of actual mouse embryos. "We found that all essential marker genes were activated at the right time in the right places in the embryoids, with only a small number of genes being out of line," says Bolondi.
When the scientists added a mutation that causes developmental defects to the model, they were able to mimic what's seen in live embryos that carry the mutation.
Sources: AAAS/Eurekalert! via Max-Planck-Gesellschaft, Science