Pencil-Eraser Tool Can Selectively Modify Proteins
Carbohydrates are essential sources of energy, but sugar molecules serve a variety of other biological functions as well. Some sugars that sit on cell surfaces function in growth or signaling, for example. They can also play a role in disease; these sugars are involved in host-pathogen interactions, inflammation, cancer, and other processes.
Sugar may now be a way to target elusive proteins that are involved in disease. While about 85 percent of proteins can't be targeted with current drugs, many contain a sugar group called O-GlcNAc (pronounced o-glick-nack). In the cell, these groups can be added to certain amino acids in a protein after it's made (a post-translational modification), and they have a variety of functional impacts. O-GlcNAc residues are found on over 5,000 proteins, many of which are now considered un-targetable with current strategies.
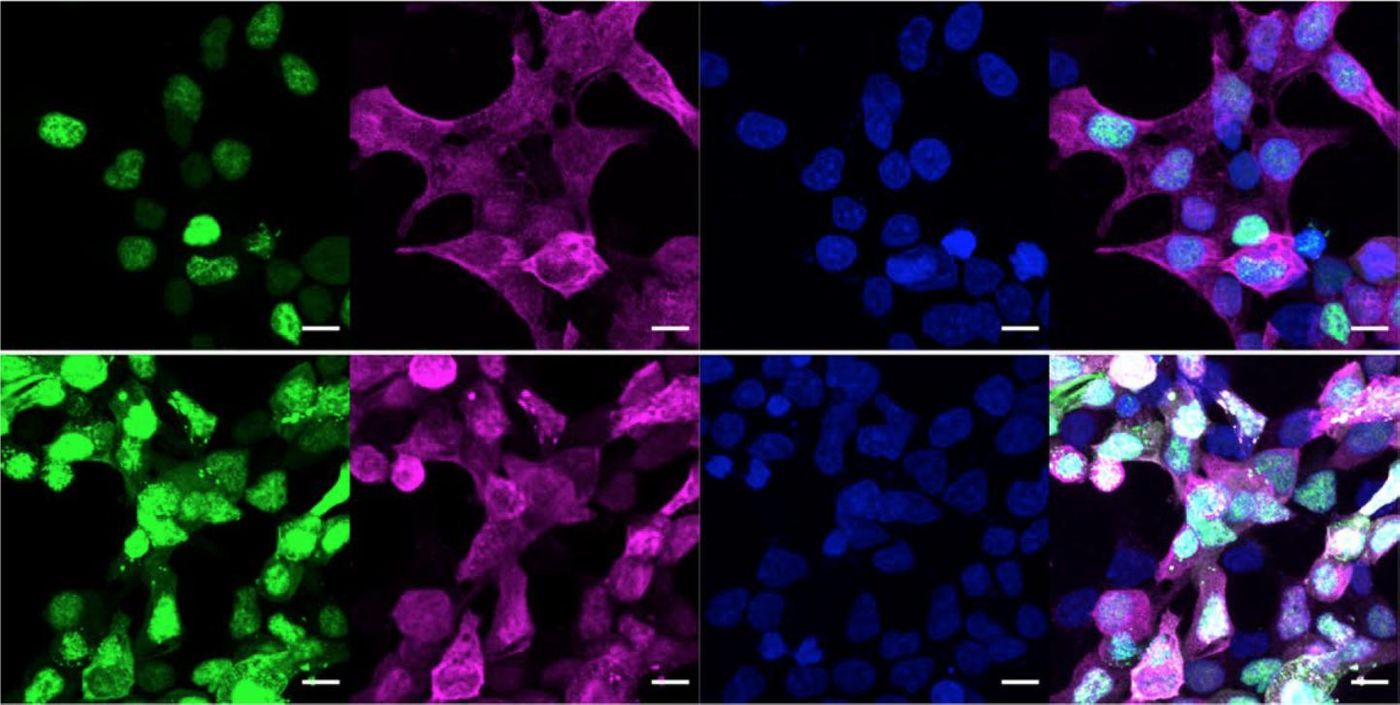
However, scientists have now created a tool that acts as a selective O-GlcNAc pencil and eraser; it adds or removes the sugar from a protein without other effects. Doing so can tell researchers about the roles of these sugars and how they might be useful in the creation of new medications. The work has been reported in Nature Chemical Biology.
"We can now start studying particular proteins and see what happens when you add or remove the sugar," explained study co-author Daniel Ramirez, a graduate candidate in biological and biomedical sciences in the Harvard University Graduate School of Arts and Sciences. "This is turning out to be very important for a lot of chronic diseases like cancer and diabetes and Alzheimer's."
The original O-GlcNAc pencil was designed by Ramirez and reported in ACS Chemical Biology. These tools find their target proteins with a nanobody, and while they can add or remove or add O-GlcNAc from a protein, they are not meant to have any other biological effect.
Current approaches enable researchers to eliminate all the O-GlcNAc in a cell, or focus in at the level of s single sugar attached to one of a protein's amino acids. Now scientists will be able to formulate more general questions about the effects of O-GlcNAc molecules on a whole protein, and how O-GlcNAc relates to disease. When the body is unable to attach O-GlcNAc to proteins, it can cause major disruptions in cells.
"With the protein-level approach, we're filling in an important piece that was missing," said study leader Christina Woo, an associate professor of chemistry and chemical biology.
The pencil-eraser tool is already in use; one group is studying the effect of O-GlcNAc on an Alzheimer's-related protein in fruit flies.
"Once you have any protein of interest, you can apply this tool on that protein and look at the outcomes directly," said first study author and postdoctoral scholar Yun Ge.
The research team wants to improve the tool more by adding optogenetic controls; that would allow scientists to turn sugars on and off by just shining a light on a cell. They also want to make an undo tool for the eraser, and add nanobodies that will zero in on specific proteins.
"We're basically trying to make the system more natural and function the way the cell does," said Ramirez.
"We really don't know what people are going to find once we give them these tools," said Ramirez. The tool may be new, but the potential is great: "We're on the iPhone one, basically, but we're already working on the next couple generations."
Sources: AAAS/Eurekalert! via Harvard University, Nature Chemical Biology