A Potential Weakness in SARS-CoV-2 is Caught on Video
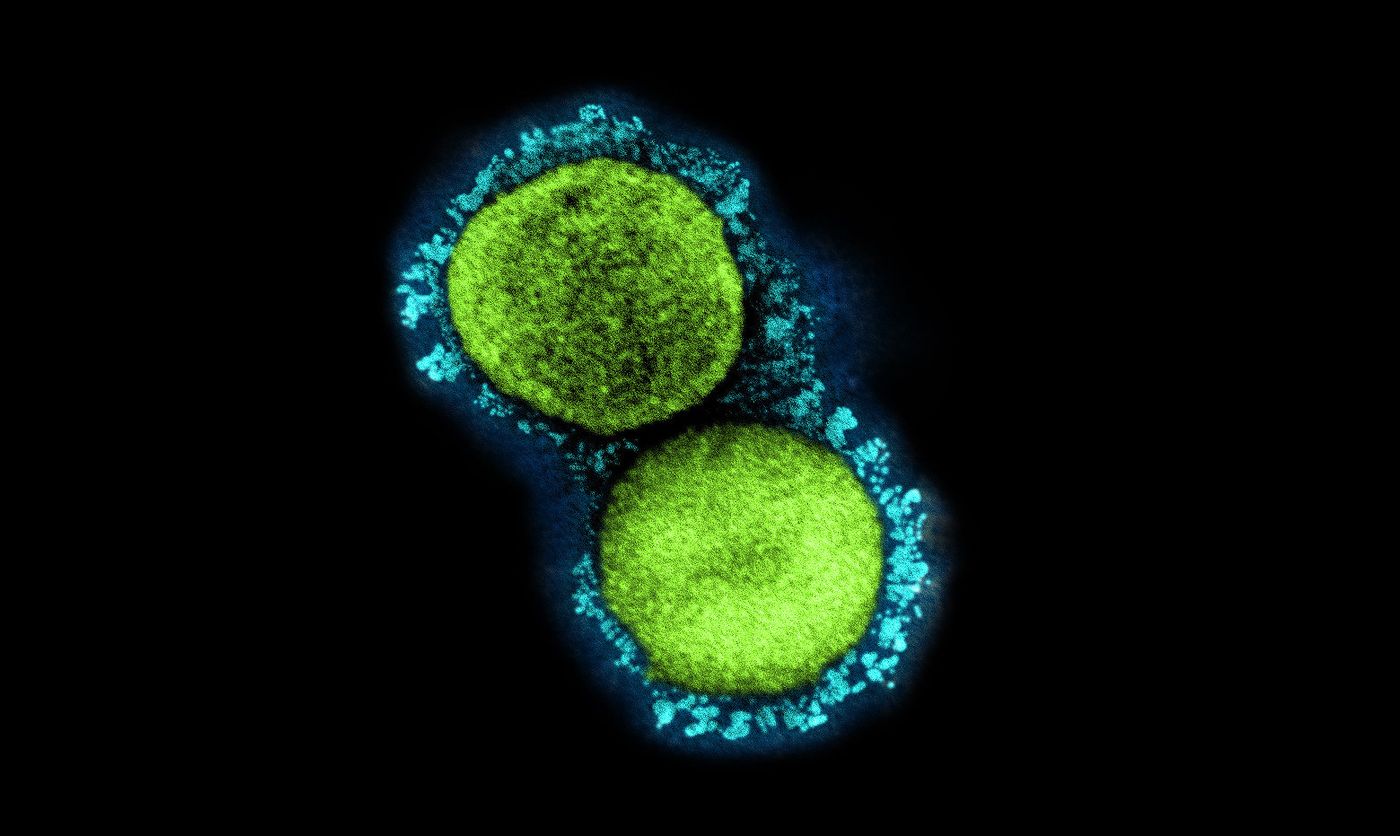
Viruses like SARS-CoV-2 have to get into host cells in order to cause an infection, and they do so by using their spike protein to bind to proteins on host cell surfaces. The spike protein of SARS-CoV-2 has been a target of much research, and we know that it can bind to several receptors including one called ACE2. Other molecules can bind to the viral spike protein too, like neutralizing antibodies that prevent the virus from causing infection. New work has demonstrated that proteins called lectins can also bind to the SARS-CoV-2 spike protein. This research, which has not yet been peer-reviewed but is available on bioRxiv, also captured that binding on video.
"Lectins are proteins that can bind the sugar molecule structure on lipids or proteins such as the spike protein," explained Dr. Josef Penninger, director of the Life Sciences Institute in Vancouver. "Our idea is to harness this property to develop a drug to combat COVID-19 disease."
Researchers have been searching for molecules that can bind to the SARS-CoV-2 spike protein. Penninger's team created a lectin library, and used it to find two lectins that are good at binding to sugars called glycans on the SARS-CoV-2 spike protein. Glycans can help disguise spike proteins, helping them to prevent detection by the immune system. Another approach is taken by the drug candidate APN01, which mimics ACE2 receptors; they are meant to compete with the receptors to bind to the virus, so the virus, in theory, would bind to APN01 instead of host cells.
"We are working with lectins on the same principle," said Penninger. "But the lectins would occupy neuralgic sites directly on the spike protein and thus disrupt the pathogen's binding to the cells. The door is blocked because the key is gummed up with lectins."
"We have now tools at hand that can bind the virus's protective layer and thereby block the virus from entering cells," said a co-first study author Dr. Stefan Mereiter. "Moreover, these glycan sites are highly conserved among all circulating SARS-CoV-2 variants, so this could be its Achilles' heel."
Study collaborator Dr. Peter Hinterdorfer and colleagues at the University of Linz measured the number of bonds and binding forces between the lectins and spike protein.
"We also filmed this bond," noted Dr. Hinterdorfer. "What is spectacular about the video is that you can see the dynamics of the spike protein."
The researchers were surprised by all the mobility in the spike protein.
"We saw that it actually opens up on the surfaces, and that the three arms are dynamic," said Hinterdorfer. "The lectins, on the other hand, were able to attach themselves to the structure for a long time on a biological scale."
Sources: Phys.org via University of British Columbia, bioRxiv