How Some Tissues 'Breathe' When There's No Oxygen
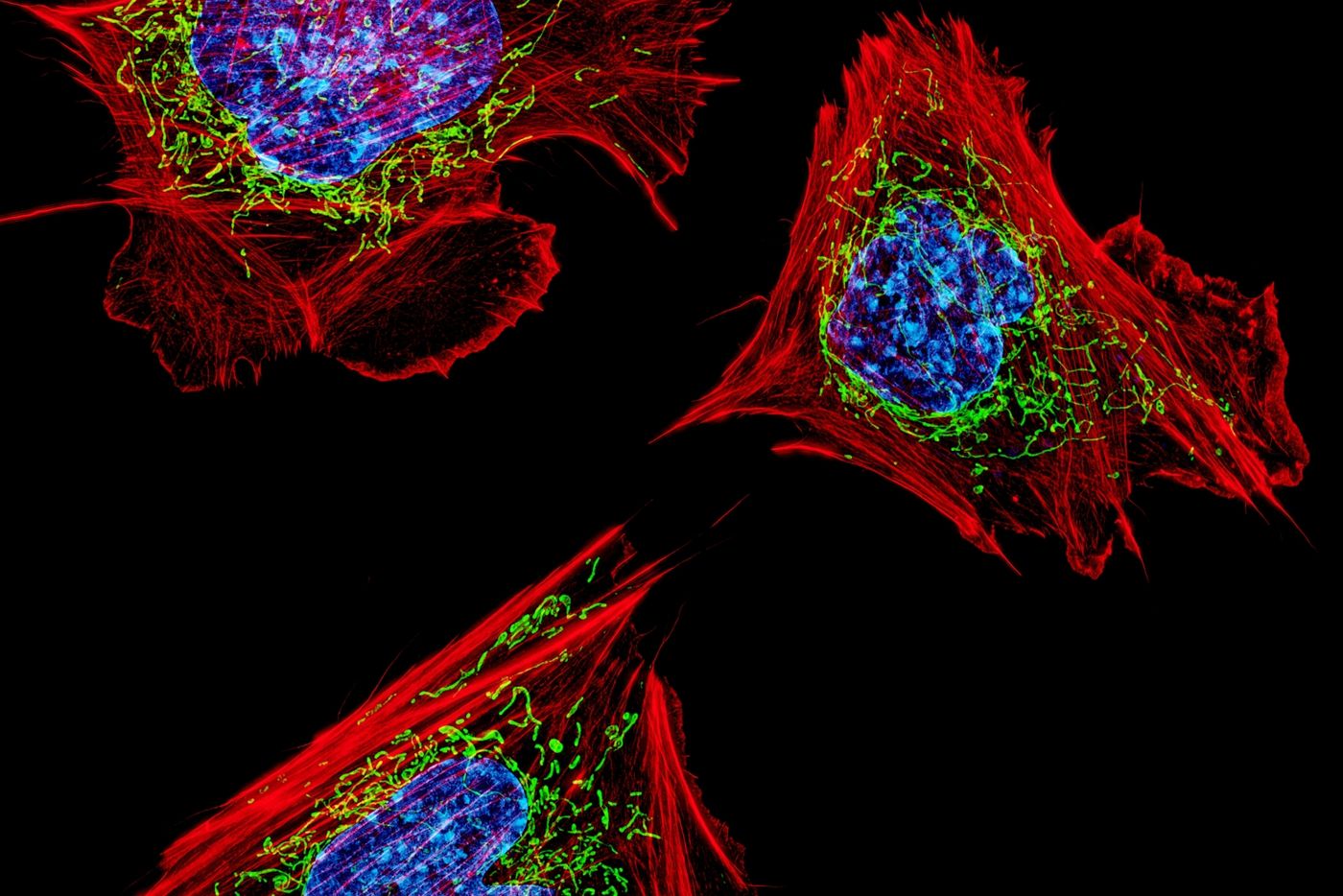
Our bodies need oxygen to survive, just like many other organisms. Oxygen molecules are used to create a molecular fuel called ATP for our cells in an organelle called the mitochondria; this process is called cellular respiration, and the electron transport chain is a crucial part of that process. At the end of the electron transport chain, two electrons are leftover, and they are coupled with an oxygen molecule, called the terminal electron acceptor. The process can then start over and generate more ATP so our cells have energy.
But there have been cases where researchers have observed that cells can keep the electron transport chain going even without oxygen. It seemed that some mitochondria can still function partially when no oxygen was available. Scientists wanted to know how they did it, said Whitehead Institute postdoctoral researcher Jessica Spinelli. The question was, "how are mitochondria capable of maintaining these electron inputs when oxygen is not the terminal electron acceptor?"
Reporting in Science, researchers have found some answers. They determined that when cells don't have access to oxygen, there's another molecule that can do its job: fumarate. Oxygen is apparently not the only terminal oxygen acceptor in the electron transport chain. This research seems to solve a long mystery, and could provide new insights into diseases in which oxygen levels drop in cells, such as cancer, diabetes, and ischemia.
A complex series of experiments was performed for this study. Mass spectrometry was used to assess the molecules that mitochondria generate when exposed to different levels of oxygen, and that revealed that in low-oxygen environments, a lot of succinate is produced. When two electrons are added to a fumarate molecule, succinate is created.
"This led us to think that maybe this accumulation of succinate that's occurring could actually be caused by fumarate being used as an electron acceptor, and that this reaction could explain the maintenance of mitochondrial functions in hypoxia," Spinelli said.
In cells, a protein complex called SDH is known to remove two electrons from succinate to leave fumarate; it ususally works in the reverse direction. Additional work showed that the complex can also work in both directions in a cell culture model.
When the entire SDH complex was removed from cells in a low-oxygen environment, the researchers found that the electron transport chain would then stop, said Spinelli.
Once this mechanism was deciphered in cells, the researchers wanted to confirm these findings with a mouse model. This work indicated that some of the tissues in the mice could use fumarate as a terminal electron acceptor, but not all of them could. Some cells even do it normally; the brain, kidney, and liver could use oxygen or fumarate as a terminal electron acceptor under normal conditions, and if oxygen was removed, fumarate could do it alone.
Other tissues were not able to use fumarate so easily. The heart and skeletal muscle could turn to fumarate, but it did not work efficiently enough to retain normal functionin those cells.
Spinelli wants to know more about this process, and how it may be working in diseased or cancerous cells.
"Certain regions of solid tumors have very low levels of oxygen, and certain regions have high levels of oxygen," Spinelli said. "It's interesting to think about how those cells are surviving in that microenvironment; are they using fumarate as an electron acceptor to enable cell survival?"
Sources: Whitehead Institute, Science