It’s long been accepted that the human genome instructs our bodies on what proteins to make using molecules called tRNAs. They have seemed to simply be an innocuous participant in a much bigger process without much impact on what happens in a cell. The interaction of tRNA and mRNA is described in the video below.
In new
work published in the journal Cell, researchers are challenging the paradigm that tRNA has no other roles or influence on the cell. It appears as though fluctuations in the abundance of tRNAs have a big impact on everything from single cells to disease processes. “We’ve long thought of these molecules as little more than middle men participating in the process of translating proteins,” said Sohail Tavazoie, a physician and Associate Professor at Rockefeller University’s Elizabeth and Vincent Meyer Laboratory of Systems Cancer Biology.
The work has been aided by improved technology, as tRNAs have traditionally been very challenging to analyze. Authors of the paper utilized approaches from next-generation sequencing to devise a new method that allows them to look at tRNA levels in depth.
Once they found a way to measure the levels of tRNAs, the research team sampled different kinds of tumors, primary and metastatic, from breast cancer patients and compared them to breast tissue samples from healthy people.
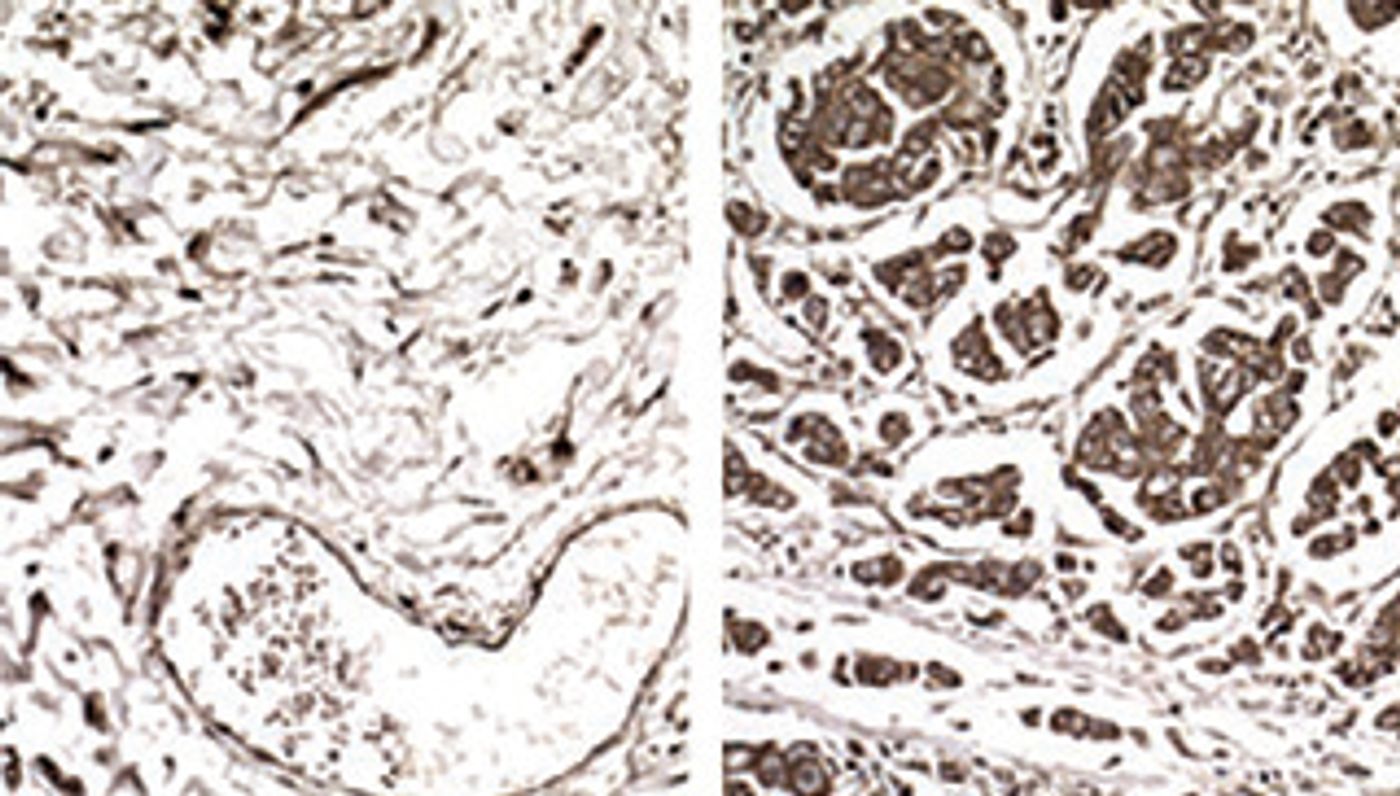
Intriguingly, it was found that the amount of two very specific tRNAs was significantly higher in metastatic tumors and cells than it was in both primary tumors that did not metastasize or healthy samples. “There are six different ways to encode for the protein building block arginine,” said Tavazoie. “Yet only one of those - the tRNA that recognizes the codon CGG - was associated with increased metastasis.” The other tRNA that was elevated in metastatic samples was one that recognizes the codon GAA and encodes for glutamic acid.
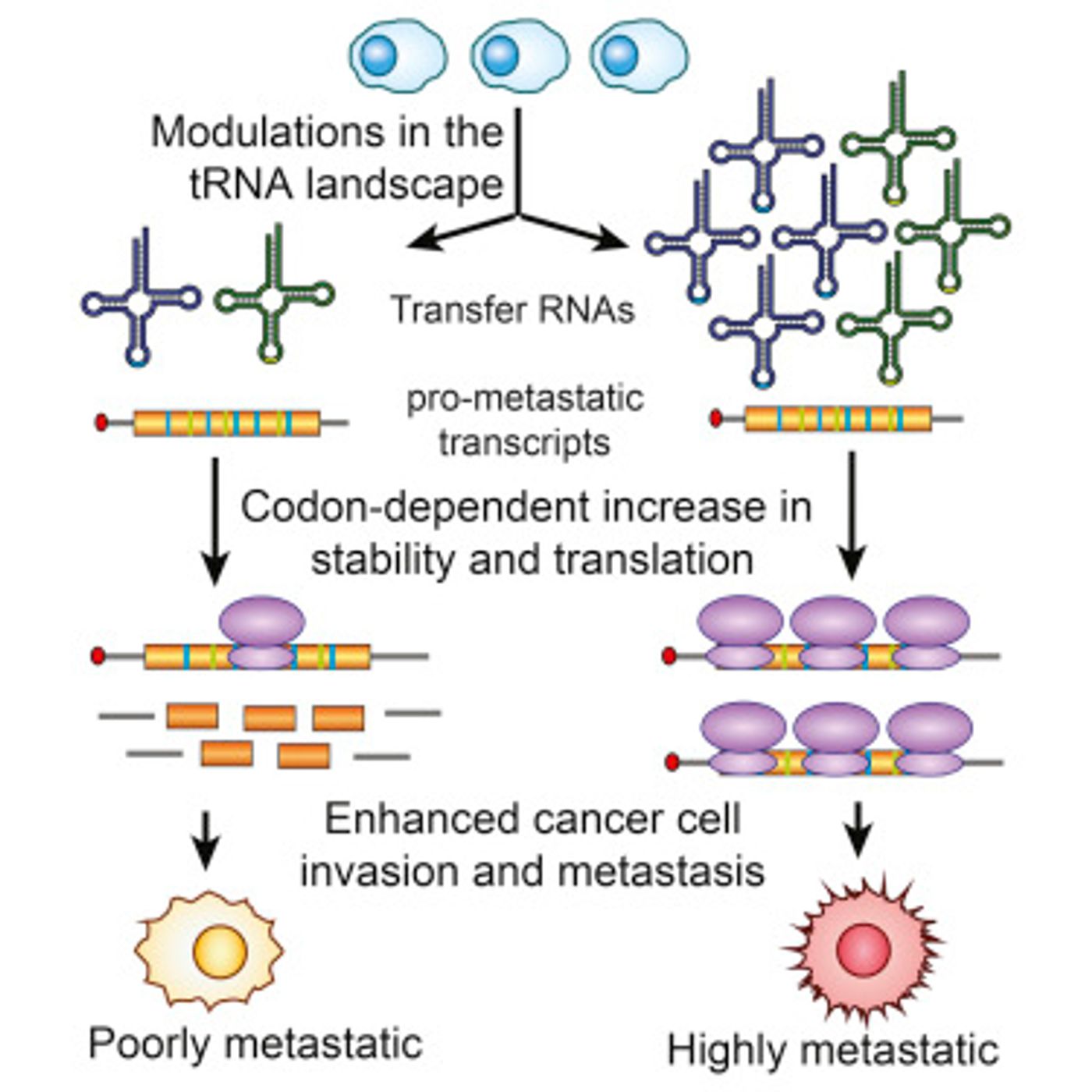
It was hypothesized that elevated levels of these tRNAs may be what drives metastasis. Using mouse models of non-metastatic primary tumors, the scientists increased the levels of two the tRNAs, and found that these cells became much more invasive and metastatic. To further validate the idea, the inverse experiment was also performed. This time, when the amount of these tRNAs was reduced in metastatic cells, a decrease in the incidence of metastases in the animals was observed, confirming their hypothesis.
So how is the tRNA causing metastasis? The team decided to look at protein levels and see how they varied when levels of the two tRNAs changed. “We found global increases in many dozens of genes, so we analyzed their sequences and found that the majority of them had significantly increased numbers of these two specific codons,” explained Tavazoie.
From their list of genes that had increased in abundance, they then altered two of them with a ’silent’ mutation. These types of mutations take advantage of the redundancy in the genetic code, and even with a single base pair change will encode for the same amino acid. When they did that, it was observed that elevating tRNA levels no longer resulted in metastasis. Tavazoie commented that “it is remarkable that within a single cell type, synonymous changes in genetic sequence can dramatically affect the levels of specific proteins, their transcripts, and the way a cell behaves.”
Sources:
Cell,
The Rockefeller University