Emergency room doctors can now diagnose a potential heart attack in 10 minutes instead of the usual hour-long wait for routine lab results. This leap in diagnostic efficiency is thanks to a new hand-held device that analyzes cardiac-specific proteins from a single drop of blood.
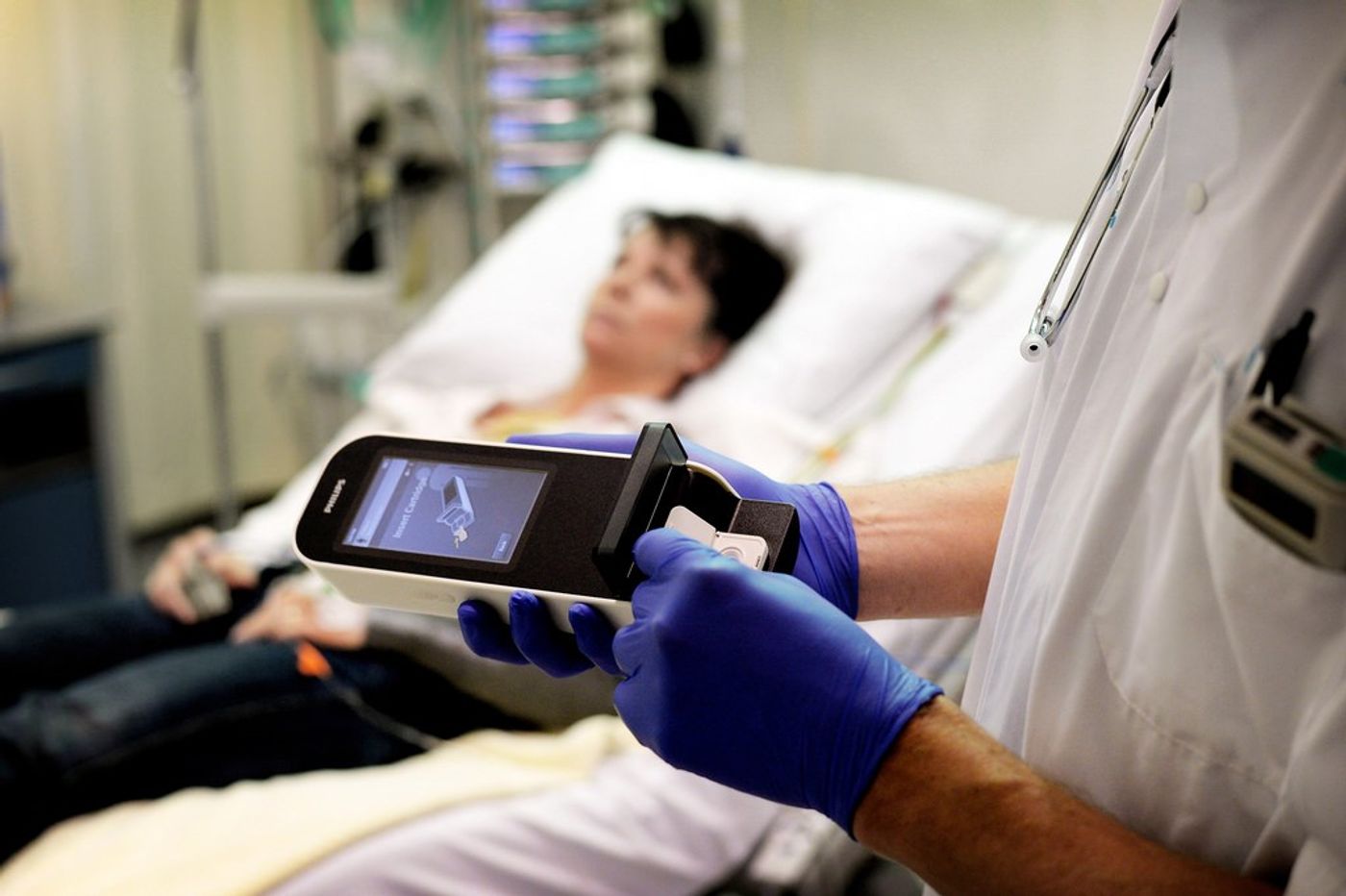
Known as the
Minicare I-20, the device resembles a common label maker. But in this portable device, Philips Healthcare has packed a quite punch. The device works by measuring the level of the biomarker cardiac troponin I (cTnI), which is a heart-specific protein that leaks into the bloodstream following a heart attack. Doctors rely on the presence of this biomarker to diagnose and confirm myocardial infarction in patients who present with chest pains. But waiting for these routine lab tests cost precious time in the ER.
"Blood samples are usually analyzed in the hospital laboratory, which can easily take more than an hour to get the result back to the ED physician. Point-of-care testing can significantly help to reduce the turnaround time," said Paul Collinson, consultant chemical pathologist at St. George's University Hospital in the United Kingdom.
With the portable device, doctors can have their patient’s biomarker results even before they complete taking the patient’s medical history. And in a setting where every minute counts, the Minicare I-20 could be life-saving, especially for high-risk cardiac patients.
"Minicare I-20 is designed to help care providers to reduce time to treatment and reduce time to discharge of patients, thereby helping to decrease crowding in the emergency department and leading to better use of hospital resources," said Marcel van Kasteel, CEO of Handheld Diagnostics at Philips.
The device passed expectations in multicenter clinical studies and in real acute hospital settings. In all validation tests, the device dramatically cuts down the diagnostic time, often exceeding a turn-around time of less than 10 minutes.
Philips has plans to extend the applications of the Minicare I-20 to other blood tests, broadening the scope of this point-of-care device. Currently, the device has not made its way to the US market yet, but it is available in the United Kingdom, Germany, and the Netherlands.
Additional source:
MNT