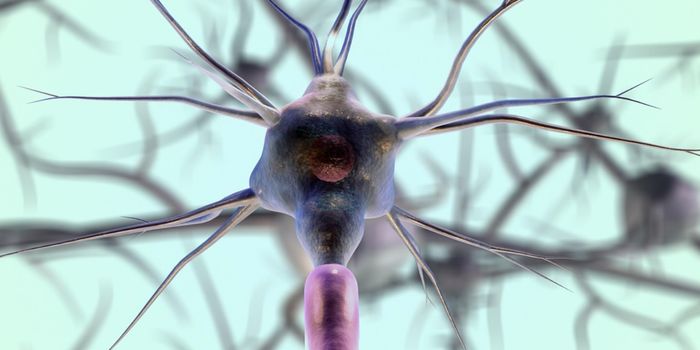
New work performed at Stanford University and published in Nature Neuroscience has revealed a neuronal circuit in the brain that is critical to the cycle of waking and sleeping. When the action of these nerve cells is suppressed in mice, those mice build nests and go to sleep. When the same circuitry was stimulated, the mice were awoken and kept up. These nerves are a component of the brain’s reward system, a complex network of nerve cells that promotes critical survival behaviors such as reproduction, in animals.
“This has potential huge clinical relevance,” explained Luis de Lecea, PhD, a Professor of Psychiatry and Behavioral Sciences at Stanford. “Insomnia, a multibillion-dollar market for pharmaceutical companies, has traditionally been treated with drugs such as benzodiazepines that nonspecifically shut down the entire brain. Now we see the possibility of developing therapies that, by narrowly targeting this newly identified circuit, could induce much higher-quality sleep.”
Dr. de Lecea reminds us that a principle question from doctors and psychiatrists to patients is, “How’s your sleep?” The National Institutes of Health estimate that some type of sleep disturbance impacts around 25 percent of American adults, with untold consequences on society.
Until this study, no exact location for the connection between arousal and reward systems had been identified. The reward system has similarities across diverse vertebrates, from fish to humans. Dopamine is one chemical implicated in the transmission of nerve impulses, such as those that go from the ventral tegmental area (VTA) of the brain to the nucleus accumbens, resulting in feelings of pleasure after achieving something or in anticipation of it. For a brief overview, check out the video below.
“Since many reward-circuit-activating drugs such as amphetamines that work by stimulating dopamine secretion also keep users awake, it’s natural to ask if dopamine plays a key role in the sleep-wake cycle as well as in reward,” explained lead author Ada Eban-Rothschild, a postdoctoral scholar. “But, in part due to existing technical limitations, earlier experimental literature has unearthed little evidence for the connection and, in fact, has suggested that this circuit probably wasn’t so important.”
The researchers utilized genetically altered mice that would allow the scientists to alter the levels of excitation of neurons in the VTA remotely. The brain activity of the mice was measured and the mice were observed on video to assess behavior.
“We knew stimulating the brain’s dopamine-related circuitry would increase goal-directed behaviors such as food- and sex-seeking” said Eban-Rothschild. “But the new study shows that at least one complex behavior is induced not by stimulating, but by inhibiting, this very circuit. Interestingly, this behavior — nest building — is essential to a mouse’s preparation for sleep.”
“This is the first finding of a sleep-preparation starter site in the brain. It’s likely we humans have one, too. If we’re disrupting this preparation by, say, reading email or playing videogames, which not only give off light but charge up our emotions and get our VTA dopaminergic circuitry going, it’s easy to see why we’re likely to have trouble falling asleep,” said de Lecea.
These findings open up the possibility for drugs that target this circuit. “We have plenty of drugs that counter dopamine,” he continued. “Perhaps giving a person the right dose, at just the right time, of a drug with just the right pharmacokinetic properties so its effect will wear off at the right time would work a lot better than bombarding the brain with benzodiazepines, such as Valium, that knock out the entire brain.”
It may be that calming this circuit can be a therapy for people with other kinds of psychiatric disorders that feature sleep disturbances, such as schizophrenia. “It could be that merely solving the sleep-wake part will clear up a lot of symptoms,” de Lecea concluded.
Sources:
Stanford News,
Nature Neuroscience