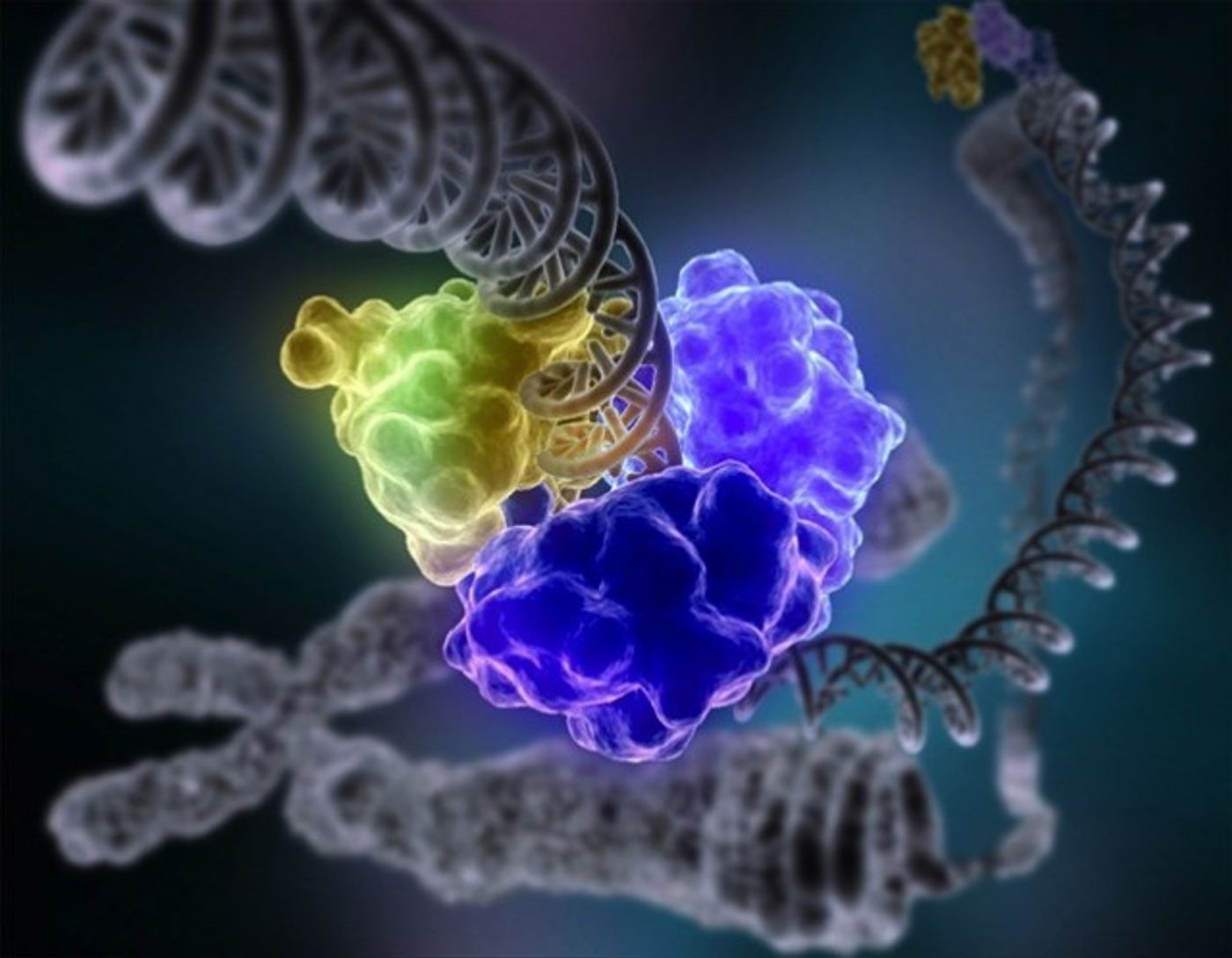
LA JOLLA, CA – September 16, 2016 – Scientists at The Scripps Research Institute (TSRI) have brought physics and biology together to further understand how cells’ crowded surfaces induce complex protein behavior.
Their findings suggest that a disordered protein, called alpha-synuclein, partially escapes from the cell membrane when it runs out of space.
“This study provides new insight into the complex structural physics of three-component protein interactions in biology,” said TSRI Associate Professor Ashok Deniz, who led the new study, which was designated as a “hot paper” by the journal
Angewandte Chemie.
A Crowded Neighborhood
Cells are crowded places, and many biophysicists have studied the effects of such crowding on the structural features of biological molecules, such as proteins. These studies tend to be done in three dimensions, with proteins suspended in a dilute solution and crowded from all sides. The new research brought scientists closer to understanding how crowding on the two-dimensional surface of a cell membrane can influence protein biophysics and function.
“This issue is particularly important given that interactions of some disordered proteins with membranes are critical elements of cellular function and health,” said Deniz.
The team focused on alpha-synuclein because it is an important example of an “intrinsically disordered protein” (IDP), which means it has relatively little structure on its own. Like several other IDPs, it can change its shape depending on interactions with nearby partners. The flexibility of IDPs makes it hard for scientists to capture clear images of them in action. Alpha-synuclein has also been linked to Parkinson’s disease, making it an interesting target for understanding the roles of IDPs in disease.
In the new study, the scientists investigated how alpha-synuclein behaves when the membrane is crowded by a second protein, called Hsp27. By adding this third component to the alpha-synuclein-membrane scene, the scientists came closer to seeing what really happens in a complex cell membrane environment, where real estate is naturally limited.
The researchers found a surprising change in the alpha-synuclein shape when it gets crowded by Hsp27. “The protein is staying partially bound, but our data suggest that part of it is flipping off the surface,” said Deniz.
The scientists said the work combined experiments, observations and simulations, which together pointed toward an explanation of the findings in terms of relatively simple physics.
Finding New Partners
The scientists think the section that detaches from the surface might work as an “arm” for waving down new potential partners.
“The fragment of alpha-synuclein can potentially interact with other binding partners, which could initiate further function,” said TSRI Graduate Student Mahdi Muhammad Moosa, who served as first author of the study with TSRI Research Associate Priya R. Banerjee.
The scientists added that other intrinsically disordered proteins may have similar reactions to crowding on membranes by proteins other than Hsp27. “We think the basic features of this model, in principle, could be applied to any intrinsically disordered protein that is bound to the cell membrane,” said Banerjee.
The researchers said the next step in this research will be to study how cell membrane composition and other modifications might alter how crowded membranes modulate the shape and cellular function of intrinsically disordered proteins.
The study, “Two-dimensional crowding uncovers a hidden conformation of α-synuclein,” was supported by the National Institutes of Health’s National Institute of General Medical Sciences (grant RO1 GM066833) and a postdoctoral fellowship from the American Heart Association (grant 15POST22520013).
This article was originally published on
scripps.edu.