Benign prostatic hyperplasia (BPH) affects nearly half of all men aged 51 - 60 and 90 percent of men older than 80 years of age. The cause of this non-cancerous enlargement of the prostate is still not conclusively known. New work done at Baylor College of Medicine and
reported in Molecular Cell reveals a mechanism for the development of the disorder. The investigators also suggest ways that current therapeutics for the disease, that target androgen, can be improved. Such a treatment strategy is not only used for BPH but also prostate cancer. If you’d like to learn more about the disease, watch the brief video below.
"Scientists have considered that inflammation can cause BPH. But how inflammation initiates in human prostate tissues and promotes BPH remains unclear," said Dr. Li Xin, Associate Professor of Molecular and Cellular Biology at Baylor and senior author of the work. "Our study provides molecular insights to answer these questions."
Initially, "we were investigating the role of the androgen receptor in prostate epithelial cell homeostasis, or how these cells balance their functions to work properly," explained Dr. Boyu Zhang, postdoctoral associate in Xin's lab and study first author.
The team wanted to understand the role of the androgen receptor in prostate cancer. Their strategy involved eliminating the receptor from luminal epithelial cells, which require androgen for survival. Other work that had been done in this area was not rigorous enough to satisfy these scientists.
"There are two limitations to those previous studies," explained Xin. "First, their approach was to delete the androgen receptor in the prostate at very early stages of the animal's development, before puberty. Therefore, it is uncertain whether the outcomes of the experiment reflect the role of androgen receptor in development or the role in homeostasis. The second limitation is that the androgen receptor was deleted in almost all of the epithelial cells, including luminal cells and basal cells. This makes it difficult to determine whether the results are a direct consequence of the cells' losing their androgen receptor or an indirect outcome of the loss of the receptor."
In this work, the researchers utilized a mouse in which a clever technique in genetic engineering allowed them to delete the receptor from some but not all of the epithelial cells, using adult mice rather then ones at young ages. As such, they were able to observe a cascade of events in which cell proliferation and inflammation combine to cause BPH in the prostate.
"When we deleted the androgen receptor in a fraction of prostate epithelial cells called luminal cells, the cells produced mediators of inflammation called cytokines, which the luminal cells normally don't do. Deletion of the androgen receptor also resulted in defects in tight junctions - connections between two adjacent cells that close the space between them," Xin said. "In normal cells, tight junctions are intact and molecules, such as mediators of inflammation, cannot leak into surrounding areas. But in the luminal cells in which the androgen receptor was deleted, tight junctions were not intact and cytokines produced by the luminal cells could leak into surrounding areas."
Thos leaked cytokines that Dr. Xin mentions brought immune cells like macrophages and T cells to the area where an inflammatory response is initiated. Additional cytokines are then secreted that in turn attract an even bigger immune response, in a feedback cycle. Growth factors then stimulate proliferation of prostate cells like luminal and basal epithelial cells.
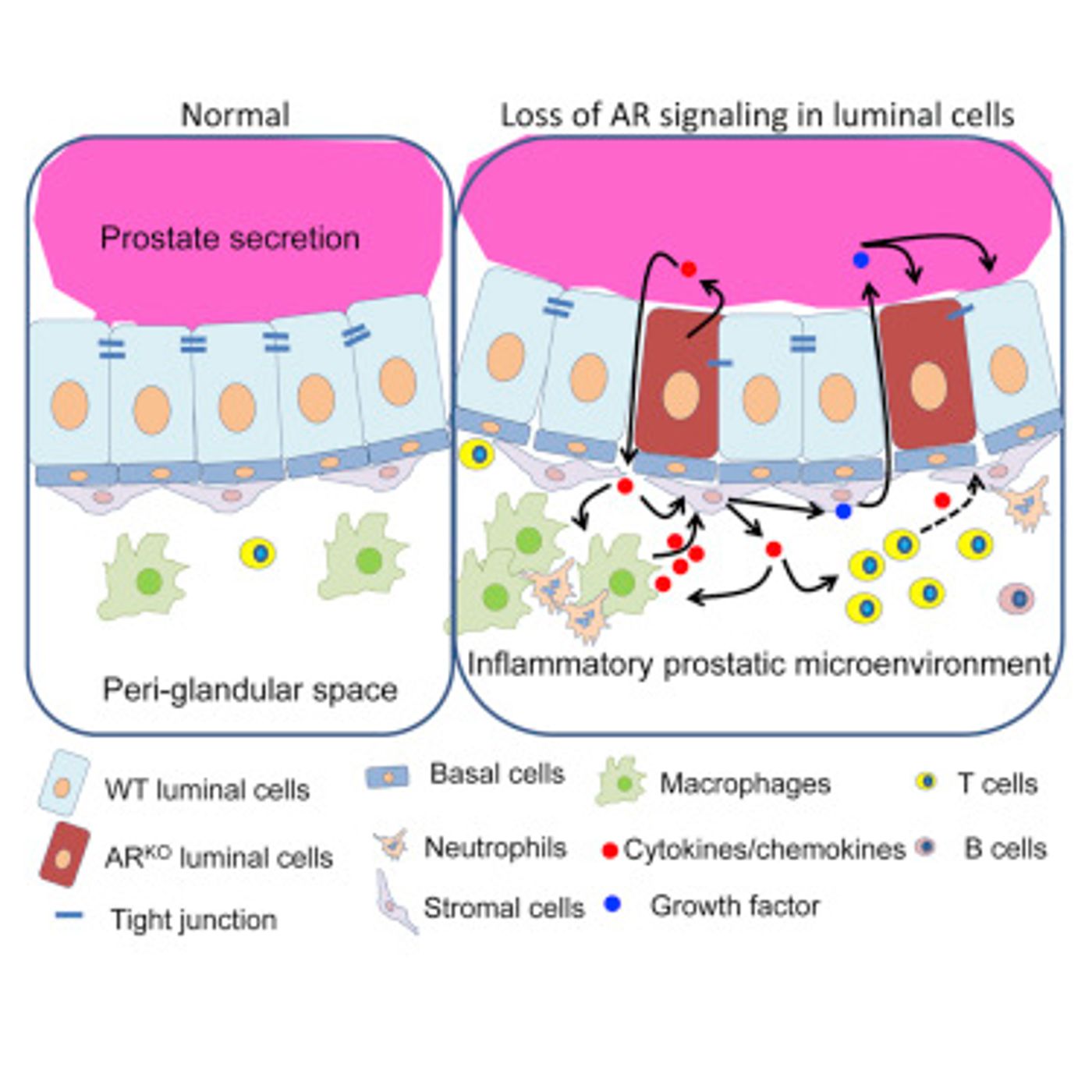
"In summary, deleting the androgen receptor triggers an inflammatory response that promotes cell proliferation," said Xin. "In the process, some luminal cells that rely on androgen signaling for their survival will die, but, at the same time, the chain of events creates a signal that promotes luminal cell proliferation. Our result that disruption of androgen receptor signaling in luminal cells can drive inflammation is consistent with the observation that luminal cells of inflamed human prostate glands have fewer androgen receptors than luminal cells from non-inflamed prostates. Our results, however, do not rule out that inflammation can affect androgen receptor expression. It is possible that this is a vicious cycle."
"We unexpectedly identified a mechanism that may explain resistance to androgen-targeting therapy," explained Zhang.
"Our results may explain why some BPH patients respond to androgen-targeting therapies, such as finasteride, while in other patients the disease continues to progress," said Xin. "We suggest that treatment for BPH might be more effective if patients received androgen-targeting therapy together with drugs that target inflammation."
This research will hopefully improve treatment options for those suffering from BPH and potentially, prostate cancer.
Sources:
AAAS/Eurekalert! via
Baylor College of Medicine,
Molecular Cell









