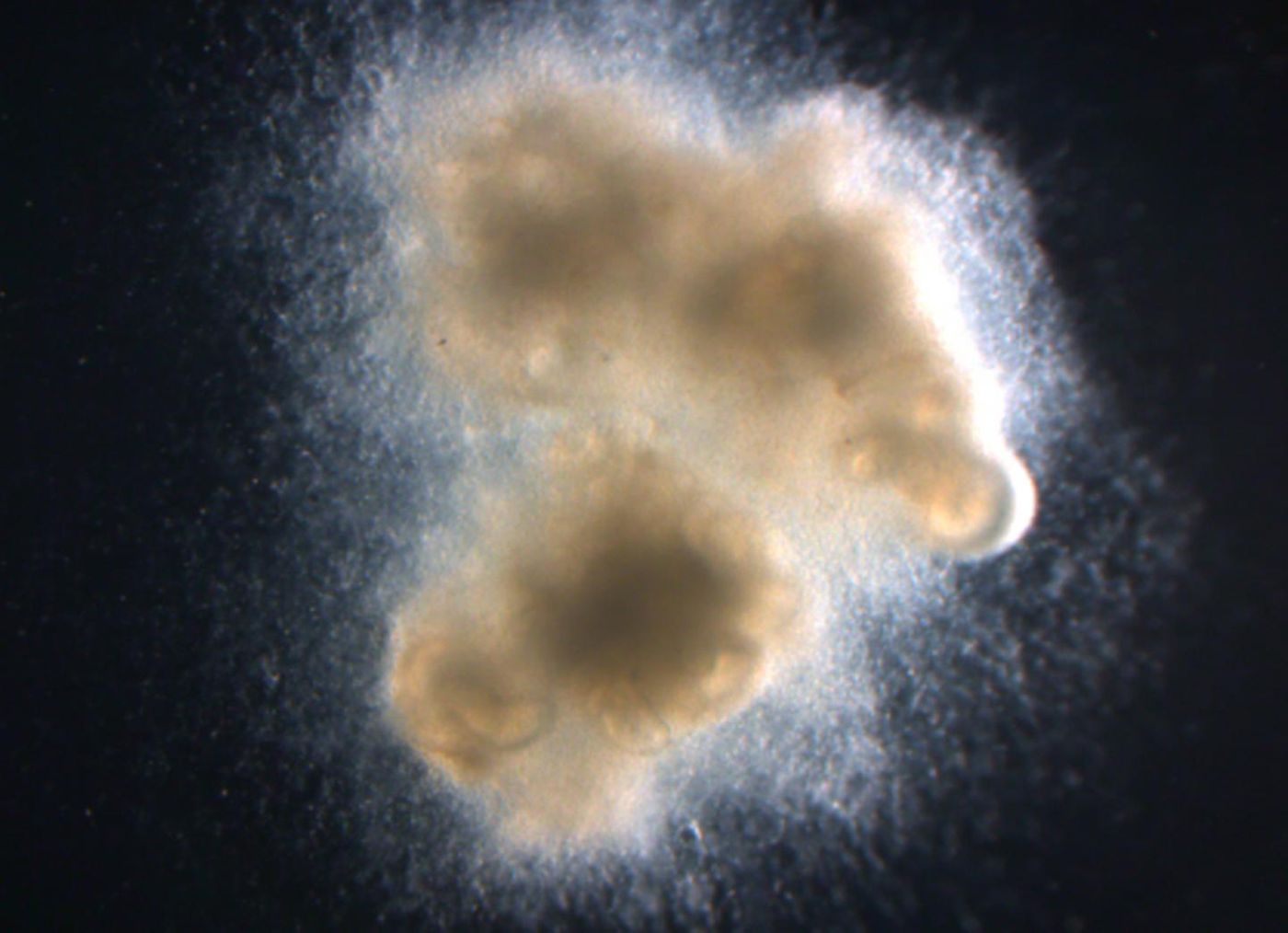
Reporting in Nature Medicine, researchers have been able to use stem cells to make intestinal organoids with functional nerves. This research paves the way for advancement in both the research and treatment of Hirschsprung’s disease, a serious intestinal nerve disorder. The authors believe the work will aid medical science in using pluripotent stem cells as a therapy in regenerative medicine and organ transplantation. The research is outlined in the following video.
"One day this technology will allow us to grow a section of healthy intestine for transplant into a patient, but the ability to use it now to test and ask countless new questions will help human health to the greatest extent," said the co-lead study investigator Michael Helmrath, MD, surgical director of the Intestinal Rehabilitation Program at Cincinnati Children's Hospital Medical Center.
The investigation of disease mechanism at the molecular level is aided tremendously by the use of a functional, three-dimensional model, especially one that can be tailored to a specific patient. This technology allows researchers to create such models from cells taken from individual patients and test therapeutics there, before taking them to clinical trials in people.
"Many oral medications give you diarrhea, cramps and impair intestinal motility. A fairly immediate goal for this technology that would help the largest number of people is as a first-pass screen for new drugs to look for off-target toxicities and prevent side effects in the intestine," explained co-lead investigator Jim Wells, PhD, director of the Pluripotent Stem Cell Facility at Cincinnati Children's.
Wells builds on his 2010
work that demonstrated the first creation of human intestinal organoids in a lab. The researchers started in the same way for this work. They added in another component, nerve cells, still at the embryonic stage, called neural crest cells. They then identified the right conditions under which the cells were to be added to the intestinal organoids in order to create an innervated nerve network.
"We tried a few different approaches largely based on the hypothesis that, if you put the right cells together at the right time in the petri dish, they'll know what do to. It was a longshot, but it worked," said Wells. The right mix resulted in intestines and enteric nerve precursor cells growing together in a way that was similar to the development of the intestine in the fetus.
To determine how these organoids grew and functioned, they had to be transplanted into a live animal; for this the researchers utilized mice with suppressed immune systems. The tissues performed very well, with robust growth, nutrient processing and peristalsis – wavy muscle contractions that push food down the digestive tract.
The researchers also used this remarkable model to study the progression of Hirschsprung's disease, a developmental disorder in which the colon and rectum don’t have a functional nervous system, resulting in the buildup of waste. When a mutation causative for Hirschsprung’s in the gene PHOX2B was used with the model, serious problems were indeed seen in the intestinal tissues.
Wells and Helmrath suggested that the manufacure of this type of functional human intestine in the lab opens up an array of new opportunities. It will now be possible to conduct more in-depth research of many subjects like nutritional health, diabetes, severe intestinal diseases, and the biochemical consequences of gastric bypass surgery.
Sources:
AAAS/Eurekalert! via
Cincinnati Children's Hospital Medical Center,
Nature Medicine