Fat Cells Help Wounds Heal as Healthy Skin Instead of Scars
In work that could be a massive breakthrough in dermatology, researchers have developed a way to coax wounds into regenerating skin instead of scarring. Something previously thought to be impossible for human skin; this process turns the most common cell type comprising wounds into fat cells. This research was performed over several years by collaborators at the Perelman School of Medicine at the University of Pennsylvania (Penn) and the Plikus Laboratory for Developmental and Regenerative Biology at the University of California, Irvine (UCI) and has been published in Science. The research is briefly summarized in the video below.
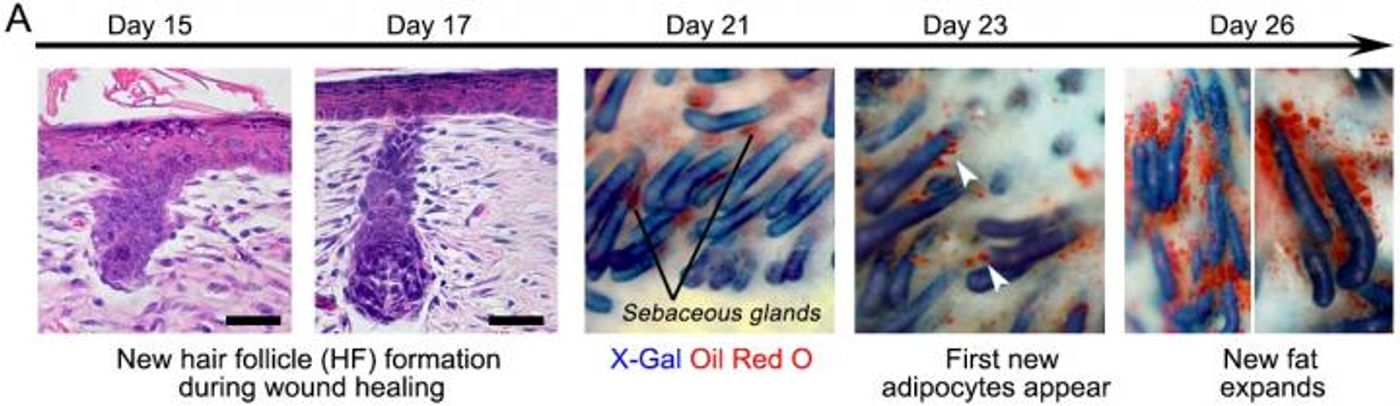
In pursuing their research, the investigators had some clues. When wounds heal they lose fat cells, called adipocytes, otherwise normally contained in healthy skin. Cells called myofibroblasts are the primary type of cell that in wounds, and it was thought that myofibroblasts could only form scar tissue. The scientists also used another characteristic of scars, the lack of hair follicles, to inform their research. Their aim was to get myofibroblasts to morph into fat cells that don’t scar.
"Essentially, we can manipulate wound healing so that it leads to skin regeneration rather than scarring," explained the principal investigator of the project, George Cotsarelis, MD, the Chair of the Department of Dermatology and the Milton Bixler Hartzell Professor of Dermatology at Penn. "The secret is to regenerate hair follicles first. After that, the fat will regenerate in response to the signals from those follicles."
Their work showed that while hair and fat form separately, they don’t develop independently. The team led by Costarelis has previously found the factors for their development. They have now found factors made by the hair follicle that convert myofibroblasts around it to form fat instead of a scar. Healed wounds just look like regular skin because they form new hairs that then stimulate the development of fat cells, which are indistinguishable from healthy cells.
Scratching beneath the surface to what was underlying the process, the researchers found that a molecule called Bone Morphogenic Protein (BMP) was the stimulation from the hair cells, signaling the myofibroblasts to turn to fat, a new discovery for myofibroblasts.
"Typically, myofibroblasts were thought to be incapable of becoming a different type of cell," Cotsarelis said. "But our work shows we have the ability to influence these cells, and that they can be efficiently and stably converted into adipocytes." The researchers demonstrated this process in human keloid and mouse cells in culture.
"The findings show we have a window of opportunity after wounding to influence the tissue to regenerate rather than scar," commented lead author of the work, Maksim Plikus, an Assistant Professor of Developmental and Cell Biology at UCI.
This work is not only applicable to wound healing. Loss of adipocytes is a problem seen in HIV treatment as well, and there is no therapy for it. "It's highly desirable from a clinical standpoint, but right now it's an unmet need,” commented Costarelis.
Aging is another condition in which adipocytes loss is a characteristic. “Our findings can potentially move us toward a new strategy to regenerate adipocytes in wrinkled skin, which could lead us to brand new anti-aging treatments,” said Cotsarelis.
As the work continues, the Plikus lab is studying other kinds of cell reprogramming and signaling in wounds beyond BMP. The Costarelis lab is examining other ways of promoting skin regeneration through its relationship to hair follicle growth.
Watch the video above, from 2014, to see a talk by Dr. Costarelis regarding hair follicles and skin regeneration.
Sources: AAAS/Eurekalert! via Penn Medicine, Science