Toward Targeted Therapeutics for Common Genetic Disorders
Research teams have made progress in the study of a group of common genetic diseases termed RASopathies because they are caused by mutations in genes that are in the RAS pathway. Two research papers were recently published, one that demonstrated a way to evaluate the effects and degree of impact of various genetic mutations, and another which could change the way RASopathies are treated. These disorders aren't well known, but they affect around one in one thousand births. They typically cause heart problems, cognitive defects, developmental delays and various facial characteristics.
"Human development is very complex and it's amazing that it goes right so often. However, there are certain cases where it does not, as with RASopathies," said co-lead author of the two studies Granton Jindal. Jindal and the other co-lead author, Yogesh Goyal, are graduate students in the lab of Stanislav Shvartsman, Professor of Chemical and Biological Engineering in the Department of Chemical and Biological Engineering at the Lewis-Sigler Institute for Integrative Genomics (LSI). "Our new studies are helping to explain the mechanisms underlying these disorders," Jindal said.
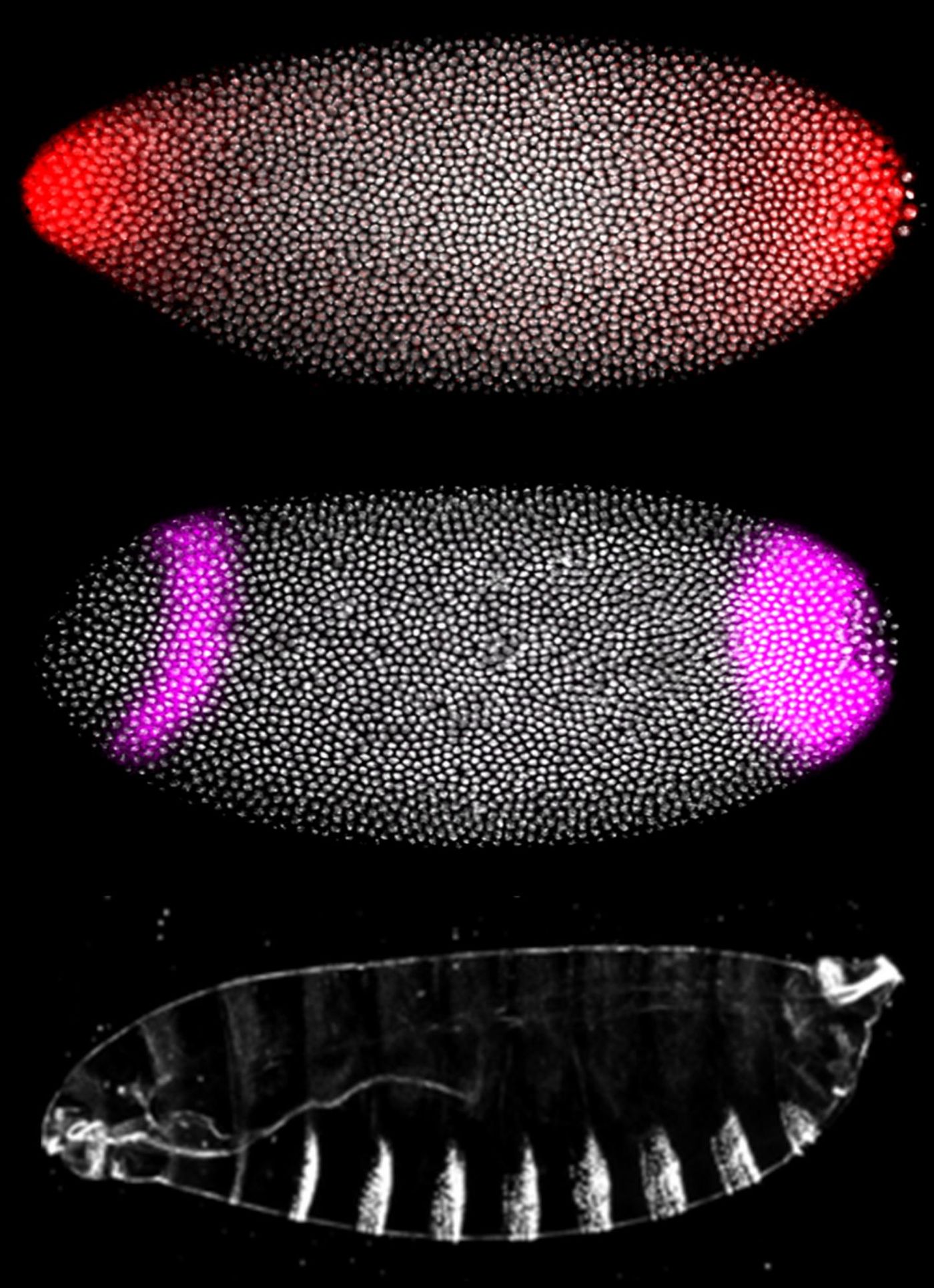
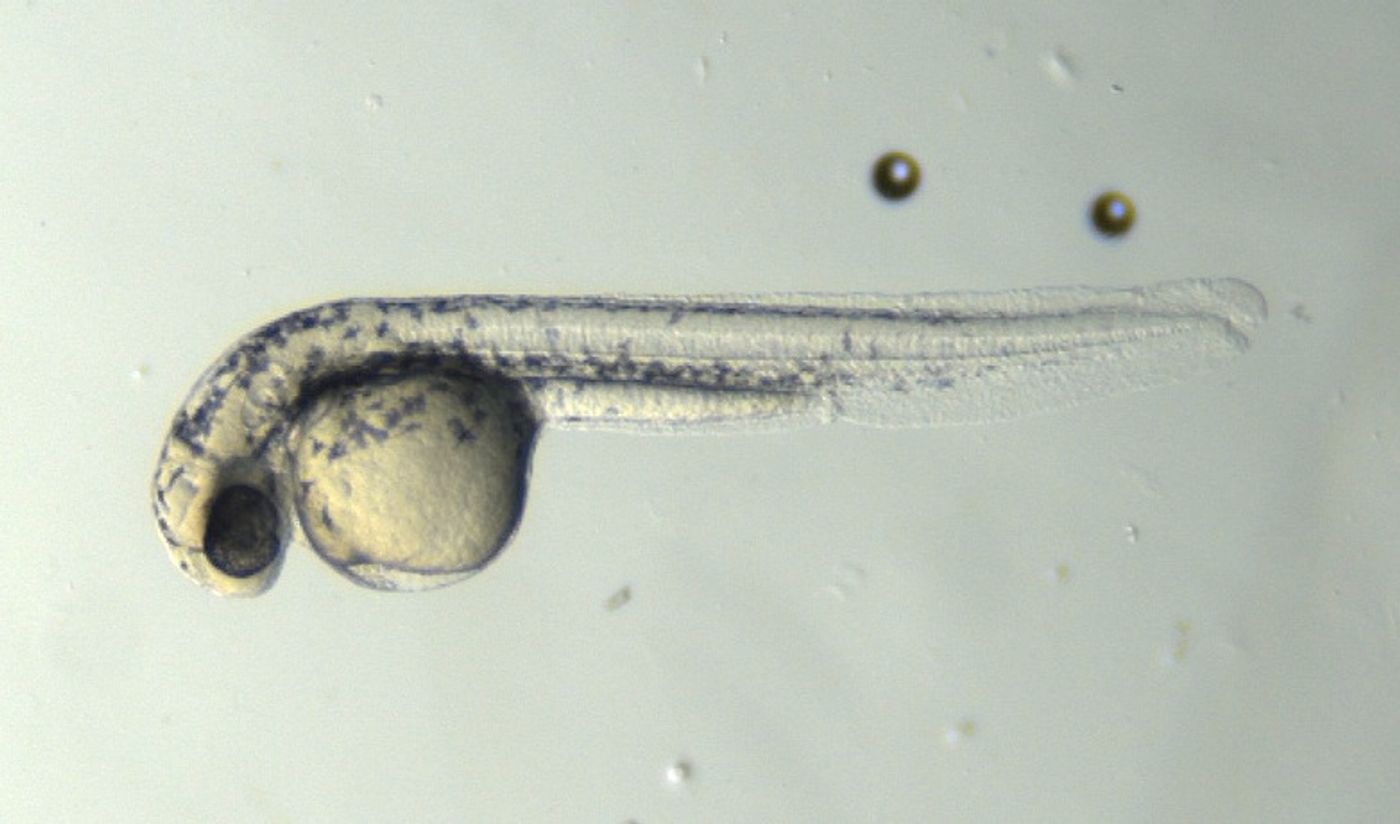
In the first study, reported in the Proceedings of the National Academy of Sciences, zebrafish embryos were used to model 16 genetic mutations individually. Zebrafish have genetic similarity to humans, and because of the speed they develop, the researchers could visualize the effects of the mutations and characterize the severity of each one. Fruit flies, another common genetic model, were also used to look at the effects of the mutations; they showed the same results.
"Until now, there was no systematic way of comparing different mutation severities for RASopathies effectively," Goyal said. The research shows how mutations might be evaluated on an individual level and could help develop better treatments for patients eventually.
"This study is an important step for personalized medicine in determining a diagnosis to a first approximation," said Jindal. The investigators also assessed the efficacy of an experimental drug as a therapeutic for those mutations. It was found that in their zebrafish model, the worse the effects from the mutation, the more drug was needed to correct the effects.
Interestingly, the second study, published in Nature Genetics, shows that some RASopathy patients might not be getting the treatment they need. RAS is one of many biochemical cascades happening within cells as a network. It has been thought that when the RAS pathway is too active, these diseases or RASopathies result. The new work indicates that certain cases of RASopathy could actually be the result of insufficient RAS signaling in some areas of the body. So drugs that work by limiting RAS pathway action could be making the problem worse.
"To our knowledge, our study is the first to find lower signaling levels that correspond to a RASopathy disease," Goyal said. "Drugs under development are primarily RAS-pathway inhibitors aimed at reducing the higher activity, so maybe we need to design drugs that only target specific affected tissues, or investigate alternative, novel treatment options."
There were several new insights into the RAS pathway from that report. RAS seems to take signals from the environment in the timing of development; mutations cause the RAS pathway to activate abnormally. Development is a carefully orchestrated series of specific events, so the slightest disruption can have a serious deleterious influence; that is what happens in the RASopathies. You can see more about them in the following video.
"Our integrative approach has allowed us to make enormous progress in understanding RASopathies, some of which have just been identified in the last couple of decades," Shvartsman said. "With continued steps forward in both basic and applied science, as we've shown with our new publications, we hope to develop new ideas for understanding and treatment of a large class of developmental defects."
Sources: AAAS/Eurekalert! via Princeton University, PNAS, Nature Genetics