A Role for Peroxisomes in Stem Cell Fate
Stem cells have gained fame for their ability to become any type of cell in the body. Unlike specialized cells with very specific functions that only make more of themselves, stem cells are undifferentiated and can either make more stem cells, while remaining undifferentiated (as only stem cells do), or they can opt to differentiate into specific cell types. Because of the many potential applications for stem cells in therapeutics and research, scientists want to know all of their characteristics, including the mechanisms that determine the fate of a stem cell. Researchers at Rockefeller University have now reported new details that underlie the decision stem cells make to differentiate or to self-renew. It seems that small structures in all cells, peroxisomes, play an important role in that decision.
"In order for the body's tissues to develop properly and maintain themselves, renewal and differentiation must be carefully balanced," said the senior author of the new work, Elaine Fuchs, the Rebecca C. Lancefield Professor and Head of the Robin Chemers Neustein Laboratory of Mammalian Cell Biology and Development. She is featured in the NIHvcast above from 2014, discussing stem cells. The new work has been published in Science.
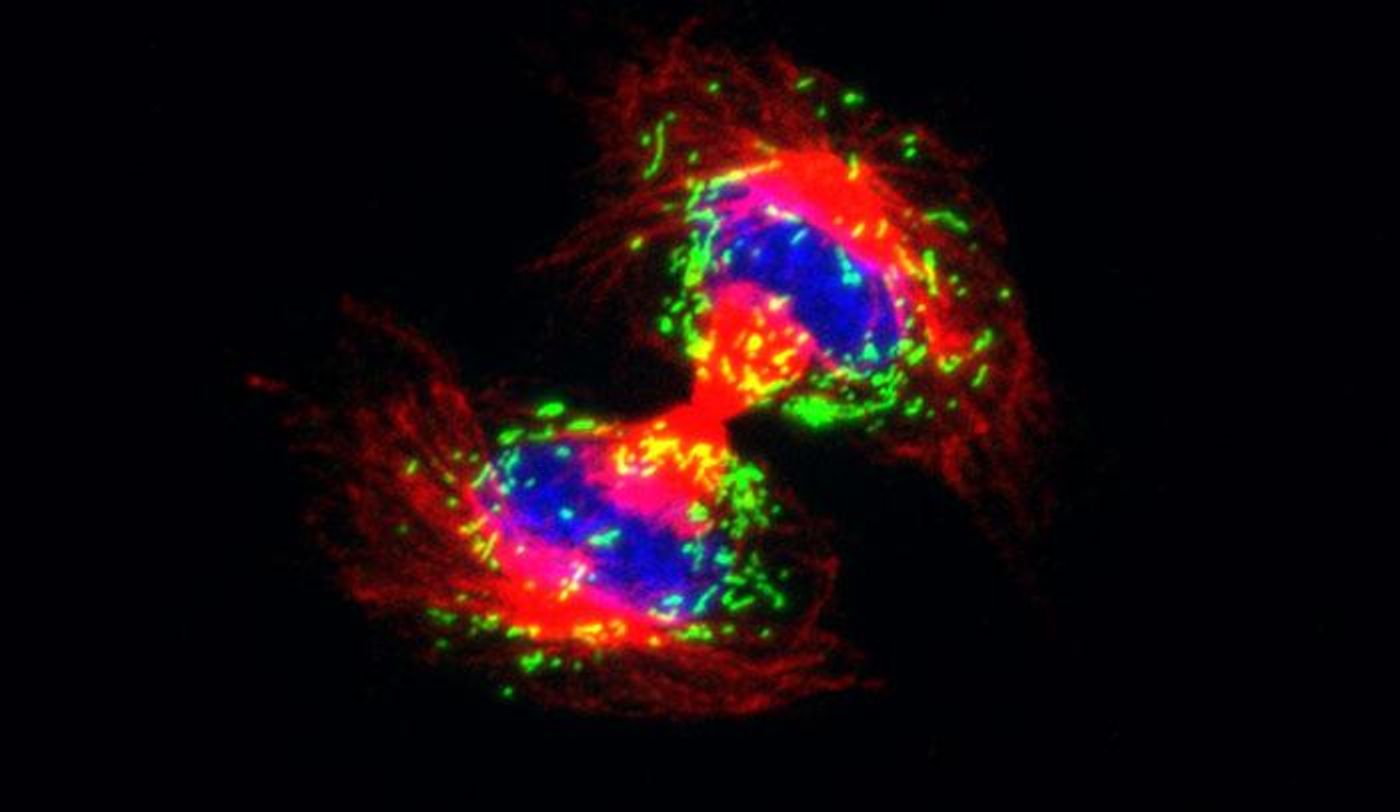
A graduate student in Fuchs' lab, Amma Asare wanted to know more about how stem cells differentiate into the epidermis, our outer layer of skin which is the main protective barrier for our bodies. Asare was able to determine that some genes play a role in that process. One gene in particular, Pex11b, is associated with the membrane of an organelle called the peroxisome. The peroxisome acts in metabolism, the breakdown of food into energy and nutrients.
The gene encodes the Pex11b protein, and Asare found that Pex11b acts to get the peroxisome into the correct place so that when a cell splits into two daughter cells, the peroxisome are evenly distributed between the two. When Pex11b was depleted from cells, the peroxisomes did not split proportionally; there were cases where all of the peroxisomes went two one of the daughter cells while the other was left without peroxisomes. In cells that were given lopsided levels of peroxisomes, cell division took longer to complete, and the architecture that separates genetic material, the mitotic spindle, was misaligned.
The net result was an abnormally low level of mature skin cells. In mouse embryos, the disruption of peroxisome location resulted in a loss of the ability to create normal skin.
"Our experiments suggest an unexpected role for the positioning and inheritance of cellular organelles, in this case enzyme-filled peroxisomes, in controlling this intricate balance," Fuchs said.
The scientists investigated the effect of physically manipulating the peroxisomes in cells, and saw the same result. "If the peroxisomes are in the wrong positions during cell division, no matter how they get there, that slows down the process," Asare explained.
"While some evidence already suggested the distribution of organelles, including energy-producing mitochondria, can influence the outcome of cell division, we have shown for the first time that this phenomenon is essential to the proper behavior of stem cells and formation of the tissue," concluded Fuchs.
You can check out a shorter video featuring Fuchs below, in which she speaks about stem cells and skin.
Sources: AAAS/Eurekalert! Via Rockefeller University, Science