Characterizing the Link Between Epilepsy and Brain Tumors
Brain tumors are difficult to detect, often only becoming apparent long after symptoms like seizures have begun. New work by researchers at Baylor College of Medicine and Texas Children's Hospital suggests that it might eventually be possible to identify brain tumors prior to the onset of symptoms, and maybe to prevent them from forming. Reporting in Nature Neuroscience, the investigators observed that when brain tumors and seizures started, a specific brain cell, one particular type of astrocyte, also started growing; there was a glaring lack of knowledge about those cells.
"We began this project by studying normal brain cells," said co-senior author of the work, Dr. Benjamin Deneen, an Associate Professor in the Center for Stem Cell and Regenerative Medicine at Baylor and the Neurological Research Institute at Texas Children's Hospital. "The brain has billions of cells of which only 30 percent are neurons. Astrocytes are the predominant cell type of the remaining 70 percent. Surprisingly, astrocytes have not been studied in as much detail as neurons have."
"Although astrocytes are often broadly categorized as one cell type, a lot of diversity exists in the functions carried out by these cells," explained co-senior author Dr. Chad Creighton, an Associate Professor of Medicine and member of the Dan L Duncan Comprehensive Cancer Center Division of Biostatistics at Baylor.
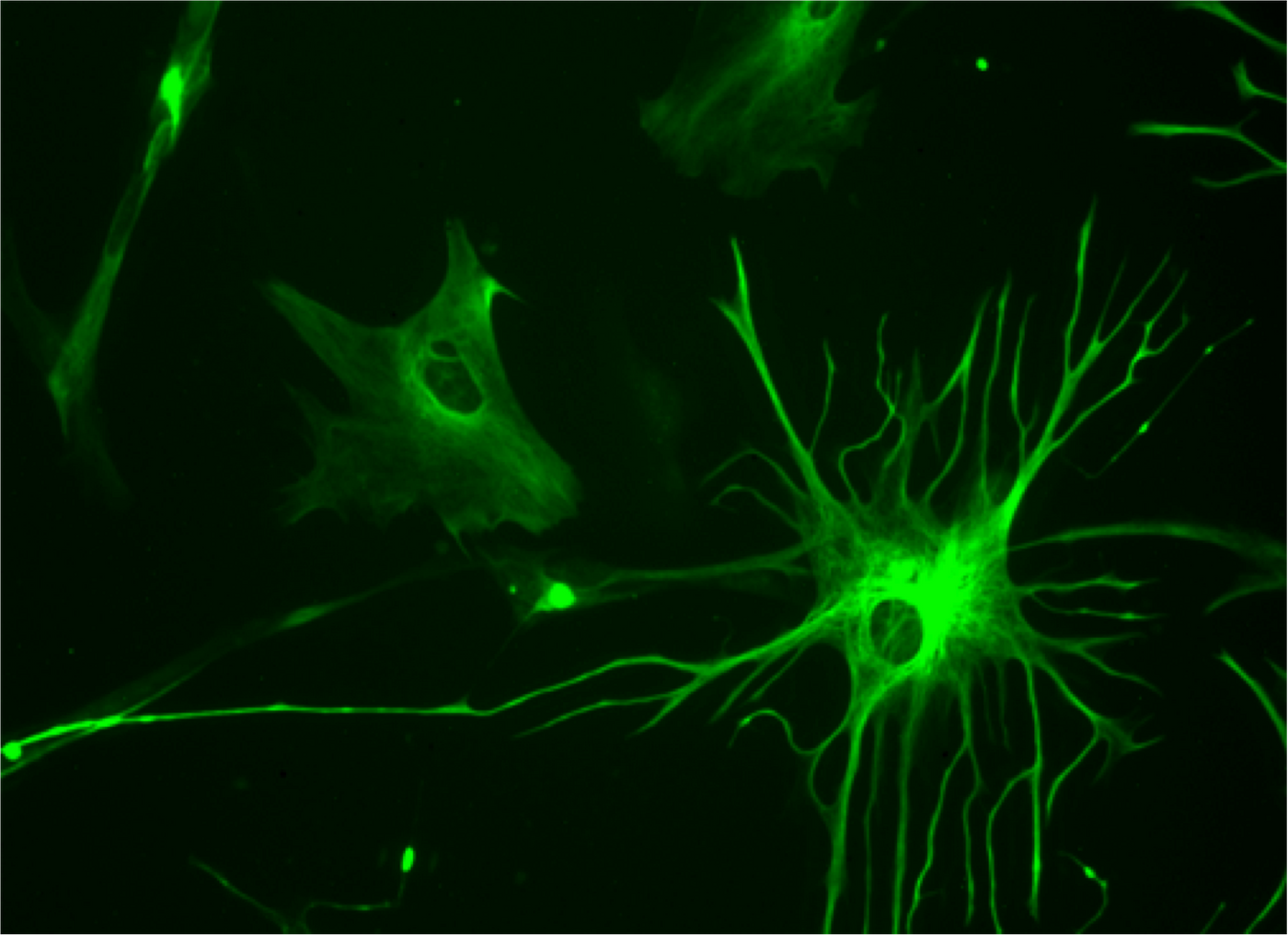
Astrocytes have many functions in the brain, such as acting to support neuronal function, aiding in neurotransmitter release and synapse formation, or creating the blood-brain barrier. They are described in the above video. Researchers wanted to find out more about astrocytes, such as whether special types carry out certain functions. To learn more, the researchers utilized molecules expressed on the surface of astrocytes as markers to separate the cells into subgroups. Five of those subpopulations were found, each carrying a specific combination of cell surface markers. These subpopulations were consistently present across various parts of the brain.
Genetic analysis revealed that astrocyte subpopulations expressed different genes, suggesting different roles for each. Genes associated with synapses were very highly expressed in one subgroup, termed subpopulation C. "In the laboratory, we combined individual subpopulation of astrocytes with neurons and measured synapse formation and function," said Deneen. "We found that neurons incubated with subpopulation C made more synapses than neurons incubated with the other subpopulations." The researchers determined that there are five different astrocyte subpopulations that uniquely support the function and formation and synapses.
"Astrocytes are associated with numerous neurological conditions, including injury, multiple sclerosis, autism, schizophrenia, Alzheimer's and Parkinson's disease and brain tumors. Given that we found diverse astrocyte subpopulations, we wondered whether these subpopulations could also explain astrocyte contributions to a host of different neurological diseases," Deneen said. His lab is researching how astrocyte development is controlled, and how astrocytes influence neurological disorders, especially the most aggressive type of brain tumor, human glioblastoma multiforme, which is described in the following video.
"We used publicly available genomic datasets to help us understand what distinguishes the different types of astrocytes from each other," said Creighton. "The genomic datasets compile entire genomes - all the genes - of different types of cells. Using this resource, we discovered that each type of human astrocyte showed very distinctive patterns of gene activation. It was by comparing these patterns with patterns associated with brain cancer or with epilepsy, using public data, that we discovered how specific types of astrocytes appear to have roles in these diseases."
The role of astrocytes in human glioblastoma formation was confirmed by two mouse models of the disease made by the researchers. Additionally, the astrocyte subpopulations were found in mouse tumors, as well as in human samples of human glioblastoma multiforme.
Epileptic seizures usually accompany glioblastomas, and are often the reason for diagnosis. The investigators at Baylor discussed their findings with a colleague, Dr. Jeffrey L. Noebels a Professor of Neurology, Neuroscience, and Molecular and Human Genetics, who confirmed that in his mouse model of brain tumors, the mice experienced seizures. They decided to investigate the astrocytes within the brain tumors of those mice; seizures correlated with the emergent growth of astrocyte subpopulation C. An increase in tumor size correlated with an excitement in nearby neurons. Additionally, astrocyte subpopulation C expresses several epilepsy-linked genes.
Other subpopulations of astrocytes, B and D, were able to migrate better in tests compared to subpopulation C. "Taken all together, the evidence from the mouse model of glioblastoma indicates that as the tumor evolves, different subpopulations of astrocyte-like cells develop within the tumor and execute distinct functions that are related to two important tumor characteristics, synaptic imbalance that can lead to seizures, and tumor migration that can lead to tumor invasion of other tissues," explained Deneen.
"Less than half of the patients with epilepsy caused by a brain tumor can be helped with existing antiepileptic drugs," said report co-author Noebels. "We do not understand exactly how malignant cells cause seizures, or why seizures persist after tumor surgery. Until now, we could only study this brain tissue at later misleading stages. I am excited that this next-generation experimental model in mice will allows us to study, for the first time, the earliest effects of human tumors on brain circuits before seizures actually begin and understand the mechanisms. These studies would be a major advance in patient care, allowing clinicians to bypass precious months spent searching for effective therapy to stop seizures. Because seizures themselves damage brain tissue, timely effective therapy is of the essence."
Sources: AAAS/Eurekalert! via Baylor College of Medicine, Nature Neuroscience