Heat-Shock Protein Chaperone Alters Environmental Effect on Mutations
The body relies on proteins for many functions; they are the essential building blocks of cells and work in maintenance, function, regulation and repair of organ systems and tissues. Strings of amino acids make up proteins, and those chains have to be properly folded into a three-dimensional shape in order to carry out their correct function. An erroneously folded protein may not only stop functioning, it could also causes serious harm. Misfolded proteins, that often end up clumping together, have been implicated in a variety of diseases. Proteins have helpers that aid this folding process, and researchers have now gained insight into other roles of those helpers.
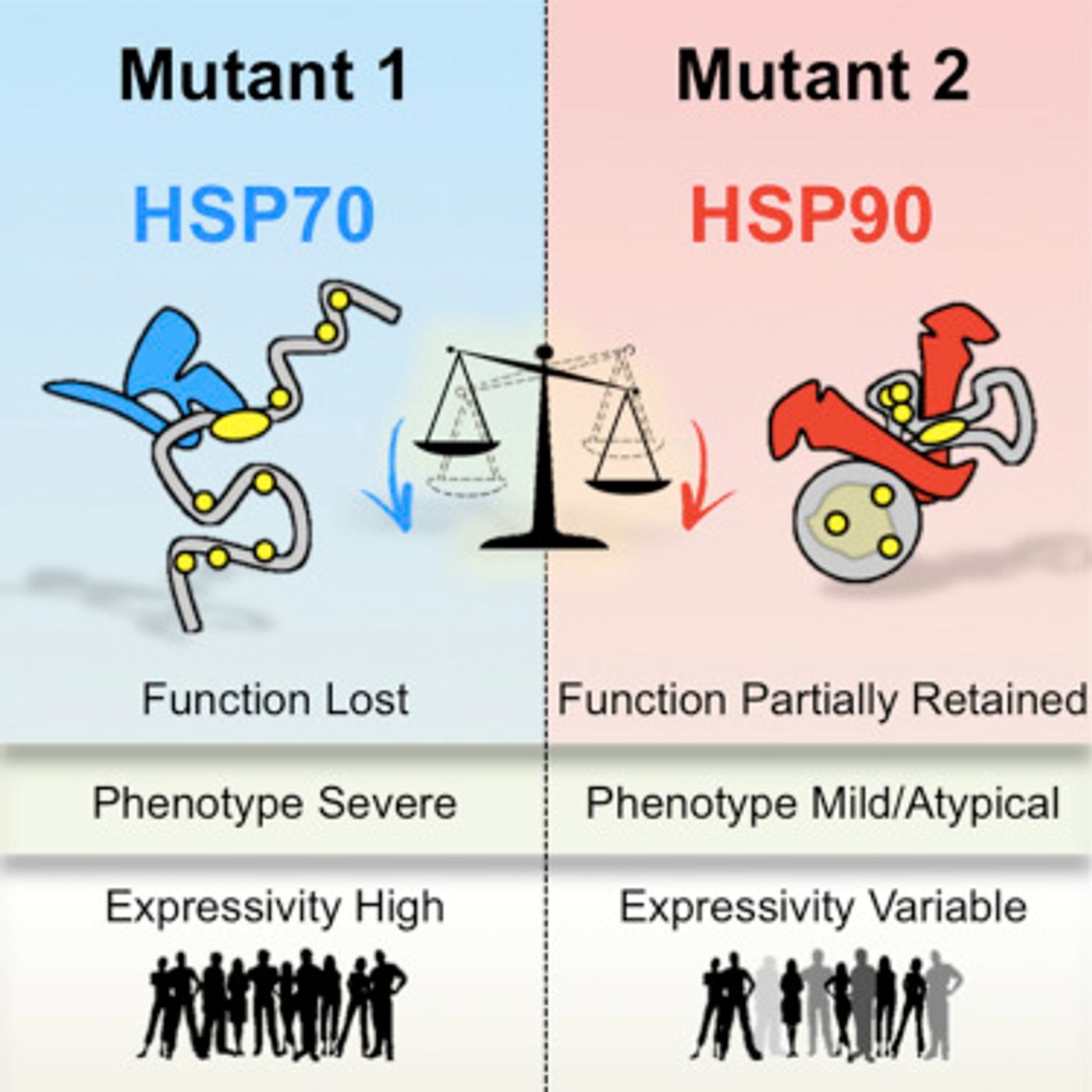
Heat-shock proteins 70 and 90 (HSP70 and HSP90) act as chaperones in the protein-folding dance, assisting a huge number of proteins as they fold into their proper conformation. It has previously been shown that these proteins can influence the impact of genetic mutations in various organisms. Reporting in Cell, Whitehead Institute scientists have now found additional evidence for HSP90 playing an important role in humans as a modifier of genetic mutation. This work has shown that HSP90 can mediate environmental effects on the function of aberrant proteins. Those influences could be strong enough to change the effect of human diseases, although that remains unknown.
"By helping proteins fold and function, HSP90 influences a range of characteristics in simple organisms on which natural selection can act, and thus alters the course of evolution," explained lead author Georgios Karras, a post-doctoral researcher at Whitehead Institute. "Our work demonstrates that HSP90 plays a similar role in humans, and in doing so it influences how disease-causing mutations manifest within cells and in the clinic. We found that HSP90 exerts a protective role on the functions of mutant proteins it directly binds, buffering the detrimental effects of mutations they carry."
HSP90 might be able to temper a mutation's effect, but that ability is very sensitive to outside influences. "HSP90 evolved to cope with environmental stressors that perturb the folding of proteins within cells," said senior author Luke Whitesell, a senior scientist at Whitehead Institute. "Yet, because HSP90's capacity is limited it can also make the mutant protein much more sensitive to environmental challenges. We observed that even mild stresses, such as fever, can provoke major effects in cells expressing HSP90-buffered mutants. This finding may help to explain why mutations can have varied clinical manifestations from person to person."
There are some genetic mutations that have more severe consequences on biological function, and they are recognized by a different chaperone molecule, HSP70. As such, the binding partner, whether it be HSP70 or HSP90, may be able to indicate the degree of severity of a mutation.
"This pattern fits well with the biochemical roles of HSP90 and HSP70 during protein-folding. HSP70 binds extended stretches of amino acids that are typically hidden in the core of the normally folded protein, while HSP90 binds to partially folded proteins. A severe mutation that leads to major unfolding and loss-of-function mainly drives HSP70 binding greater than HSP90 binding. Milder mutations have the opposite effect. Based on this pattern, we estimate that HSP90 can influence the consequences of up to 25 percent of missense mutations in some genes," Karras explained.
This work utilized data on over 1,500 disease-causing mutations that have been associated with myriad diseases. The work could eventually aid in the clinic as doctors work to find a more specialized treatment for various dysfunctions in protein folding, and could help explain clinical variability in genetic diseases.
Learn more about heat shock proteins in the following video from Shomu's Biology.
The video below features another Whitehead Institute researcher, Susan Lundquist, presenting a talk on how protein folding is related to human disease.
Sources: AAAS/Eurekalert! via Whitehead Institute, Cell