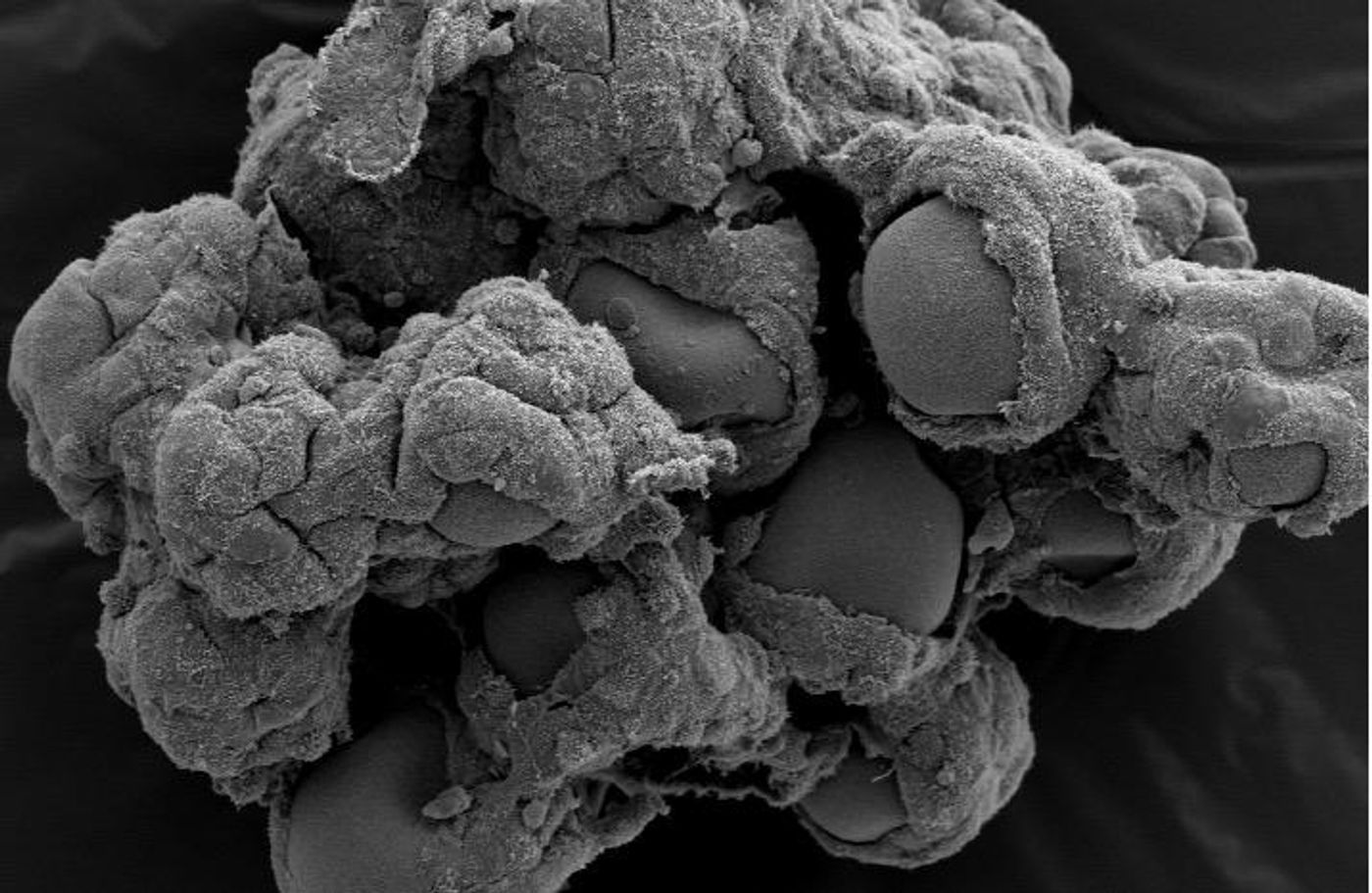
3D Model of the gut Created for Infectious Disease Research
Even with the power of medicines like antimicrobials and preventative measures like vaccines, infectious diseases remain the leading cause of death in the world among kids and young adults. Developing and testing new therapeutics will be essential to address current and future threats to public health, and that will require reliable models for research use. While cell culture models are good, they don't often mimic the three-dimensional environment of tissues; as such, their correlation to the reality of human physiology is sometimes questionable. A new tool has been created by researchers at the Biodesign Institute of Arizona State University that models the intestine in three dimensions. The work results from a collaborative effort of ASU and NASA's Johnson Space Center, and was led by Cheryl Nickerson, a Professor in the School of Life Sciences of ASU. The research team wants to engineer these intestinal tissue models primarily for the research of leading food poisoning culprit Salmonella, which has a massive deleterious economic effect.
"We engineered our advanced 3-D co-culture model to incorporate an important immune defense cell type found in the intestine, macrophages, which are key cells targeted by Salmonella during infection and are important for its disease-causing potential," said Nickerson, who is the corresponding author of the report. "The inclusion of macrophages along with epithelial cells allows for synergistic contributions of different cell types to be evaluated during infection so that host-pathogen interactions can be studied in a more physiologically relevant context. Above you can see a video featuring Nickerson.
This research utilized Salmonella strains that result in gastroenteritis and deadly bloodstream infections, including multidrug resistant Salmonella strain D23580. That strain has been implicated in terrible outbreaks of invasive infections of the bloodstream in sub-Saharan Africa. Nickerson's team has continued their groundbreaking work, using bioreactor technology from NASA to make a complex model that more accurately reflects human tissues and connect the data obtained from animal models and traditional cell culture studies. For the first time immune cells have been utilized in a three dimensional model of the intestine created with the NASA rotating wall vessel (RWV) bioreactor, and the first time that this co-culture system has been used to study infections that impact the human gut.
"Our co-culture model thus offers a powerful new tool in understanding enteric [gut] pathogenesis and may lead to unexpected pathogenesis mechanisms and therapeutic targets that have been previously unobserved or unappreciated using other intestinal cell culture models," said Nickerson.
For a successful three dimensional model, scientists have to try to recapitulate the microenvironment of the intestine, including features like oxygen levels and physical forces generated by fluid flow. Therefore, a host-pathogen interaction is more accurately reflected. In this work, Salmonella was grown in varying oxygen levels such as those encountered in different regions of the intestine.
The dynamic bioreactor technology was utilized so cells could be grown in the same fluid shear as that in the intestinal tract.
"It's a real challenge for researchers to accurately model all of the steps involved in the initiation and progression of host-pathogen interactions in the laboratory due to all of the complex factors in the human body that contribute to infection," said one author of the work, Jennifer Barrila. "By harnessing the naturally low fluid shear environment generated by the NASA RWV bioreactor during model development combined with the use of physiologically relevant oxygen conditions during infection, we have come several steps closer to achieving our ultimate goal of recreating this complex 3-D microenvironment."
When the researchers studied the effects of various Salmonella strains in the model, strains which have very different characteristics and result in different diseases. They found that the model in turn responded very differently to each strain; there were various modes of colonization - adherence, invasion and intracellular survival - as well as different intracellular co-localization patterns in immune and epithelial cells.
"One important advantage of using this 3-D multicellular in vitro host model system is the ability to visualize the co-localization patterns of different pathogens within the different host cell types. I believe that these platforms can advance our knowledge of a variety of enteric diseases of both infectious (bacterial and viral) and non-infectious etiologies (IBS/IBD, drug toxicity, etc)," said another author of the report, Jiseon Yang.
The model has been shown to be effective and useful in the study of enteric pathogenesis. The researchers are now building upon the findings and adding layers of complexity with the ultimate goal of modeling the entire organ. They have even looked beyond that, considering how immune cells might be added to the brew to further mimic the real physiology.
"The future of this field is limitless, and we are still in the infancy of learning how to build more realistic and complex models of native human tissues to better understand host-pathogen interactions and infectious disease mechanisms. These findings are urgently needed for new vaccine and drug development to outpace infectious disease. It is thus exciting to see the infectious disease world begin to embrace 3-D tissue models." concluded Nickerson.
Sources: AAAS/Eurekalert! via ASU, NPJ Microgravity