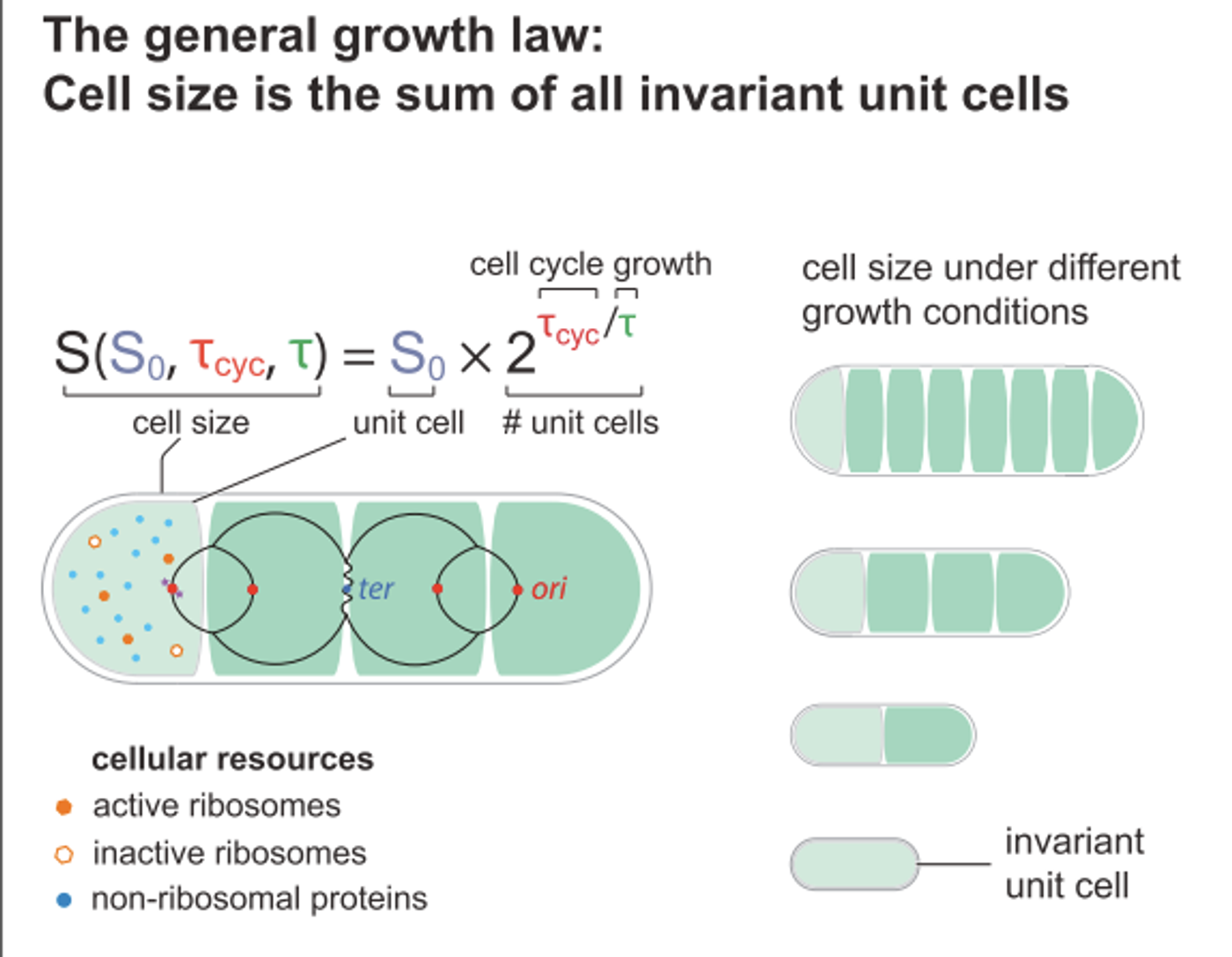
Bacterial Cells Have a Fundamental Size
The expansion of bacterial colonies depends on the availability of sustenance, and when nutrients are good quality, microbes are able to grow bigger and more quickly. There is a principle connecting the size of bacterial cells to their proliferation rate: a growth law. However, that law fails to explain why bacteria, regardless of environmental conditions, divide once they reach a particular size. New work has modified that growth law to resolve this issue.
A team of physicists, biologists and bioengineers from UC San Diego that performed the research have published their findings in the journal Current Biology.
"A few years ago, we set out to do extensive growth inhibition experiments to test the growth law using the model organism Escherichia coli," explained Suckjoon Jun, an assistant professor of physics and molecular biology at UC San Diego, who headed the research effort. "Perhaps not so surprisingly, the original growth law was unable to explain changes in cell size under growth inhibition. Cell size either increased or decreased depending on the inhibition method. Sometimes, cell size did not change at all despite significant growth inhibition."
The research team observed that when the genetic material inside of cells began to replicate in preparation for the division of a cell, the size of the cell tended to stay the same even with all of the changes that were taking place inside of it, such as the synthesis of new proteins, the production of new DNA, various alterations in cell shape and additions to the cell wall.
"We realized that this invariant cell size represents a fundamental unit of cellular resources required to start growth and the cell cycle, or the 'engine' of a car, so to speak," said Jun. "This 'unit cell' is the fundamental building block of cell size, and cell size is the sum of all invariant unit cells for any growth condition, explaining the origin of the growth law."
Jun credited the advancements of genetic manipulation techniques like the CRISPR gene-editing tool and high-throughput cell sampling tools for enabling his team to harvest a wealth of physiological data from 10 million bacterial cells that were used in their growth inhibition experiments.
"This allowed detailed and reliable statistics, and led to quantitative modeling that made experimentally testable predictions, helping us to understand the data at a deeper level," Jun added. "This complements the unexpected 'adder' principle that we discovered a few years ago."
Jun related this process to the techniques of the Danish astronomer Tycho Brahe. Four centuries ago, he was able to convince the German astronomer Johannes Kepler that planetary orbits, whose origin is gravity, were not circles but ellipses through painstaking collection of better orbital data.
"Kepler's elliptical model said nothing about the physical origins of ellipses, but his kinematic modeling was an essential starting point for Newton's work on dynamics 50 years later," Jun explained. "We don't know whether biology is following the footsteps of the history of physics, but examples are accumulating that some branches of biology are becoming as quantitative a science as physics."
You can check out a full lecture from Professor Jun on the coordination of growth and the cell cycle in the video.
Sources: AAAS/Eurekalert! via UCSD, Current Biology