New CRISPR-Gold Tool Repairs Muscular Dystrophy in Mice
Scientists at the University of California, Berkeley, have created a new way to deliver the gene-editing tool CRISPR. The CRISPR-Cas9 editing system was invented several years ago and has rapidly been tested in a variety of models. While some drawbacks have been identified, scientists have also tweaked the method to allow for more precise and efficient editing. Now, investigators report that their new delivery system, CRISPR-Gold, can repair the genetic mutation that causes Duchenne muscular dystrophy. Mice with Duchenne muscular dystrophy that received a single injection of CRISPR-Gold exhibited a two-fold increase in strength and agility and a correction rate that was 18 times higher compared to control groups.
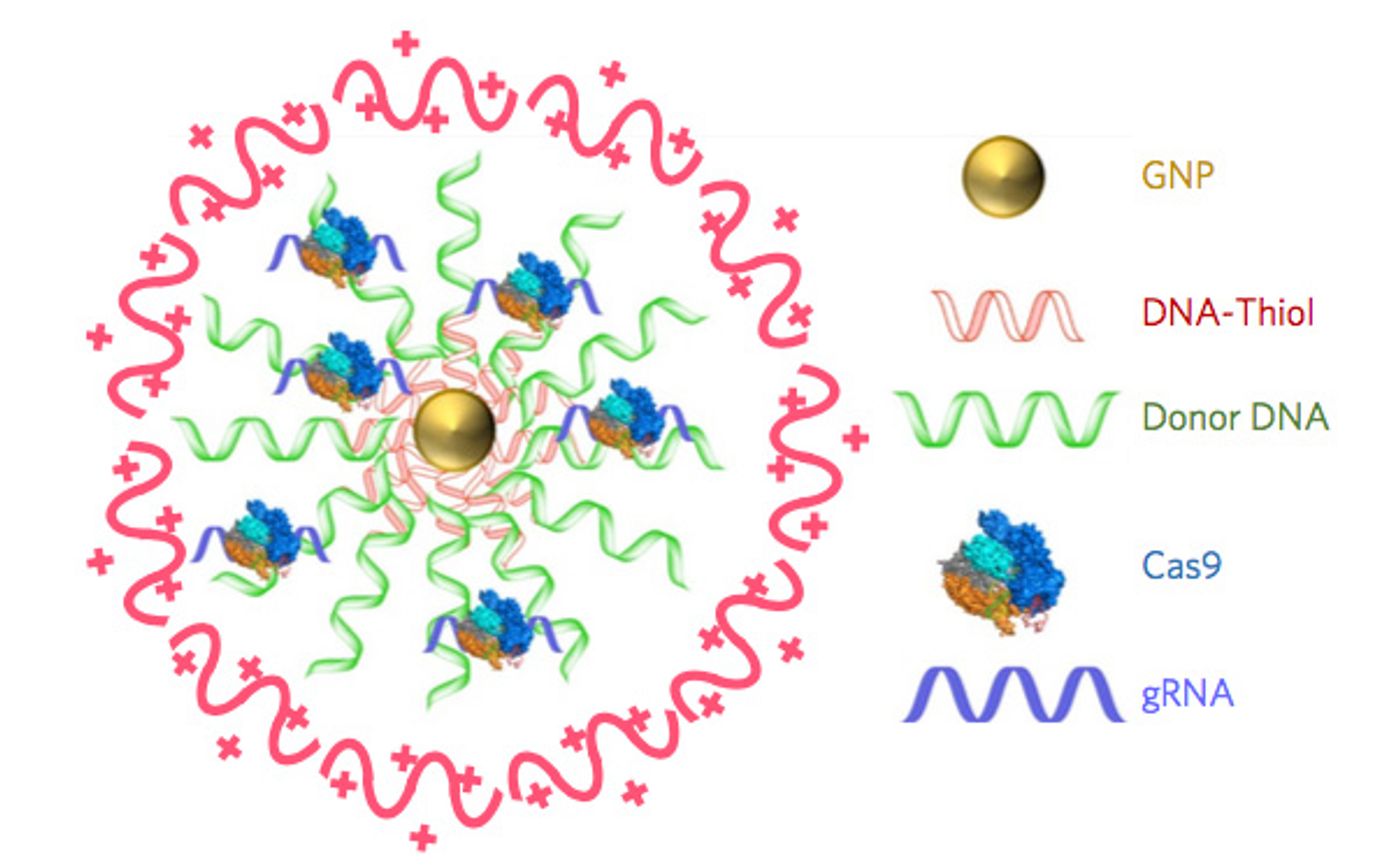
While the relatively precise gene-editing action of CRISPR-Cas9 is very attractive for researchers trying to correct errors in the genome that lead to disease, several components are needed for it to work. Getting all of that stuff into cells is challenging, and viruses are usually employed as the delivery system. These viruses can also have unintended consequences, in addition to the ones that may come from the CRISPR-Cas9 system itself. The new CRISPR-Gold tool does not require a virus, however.
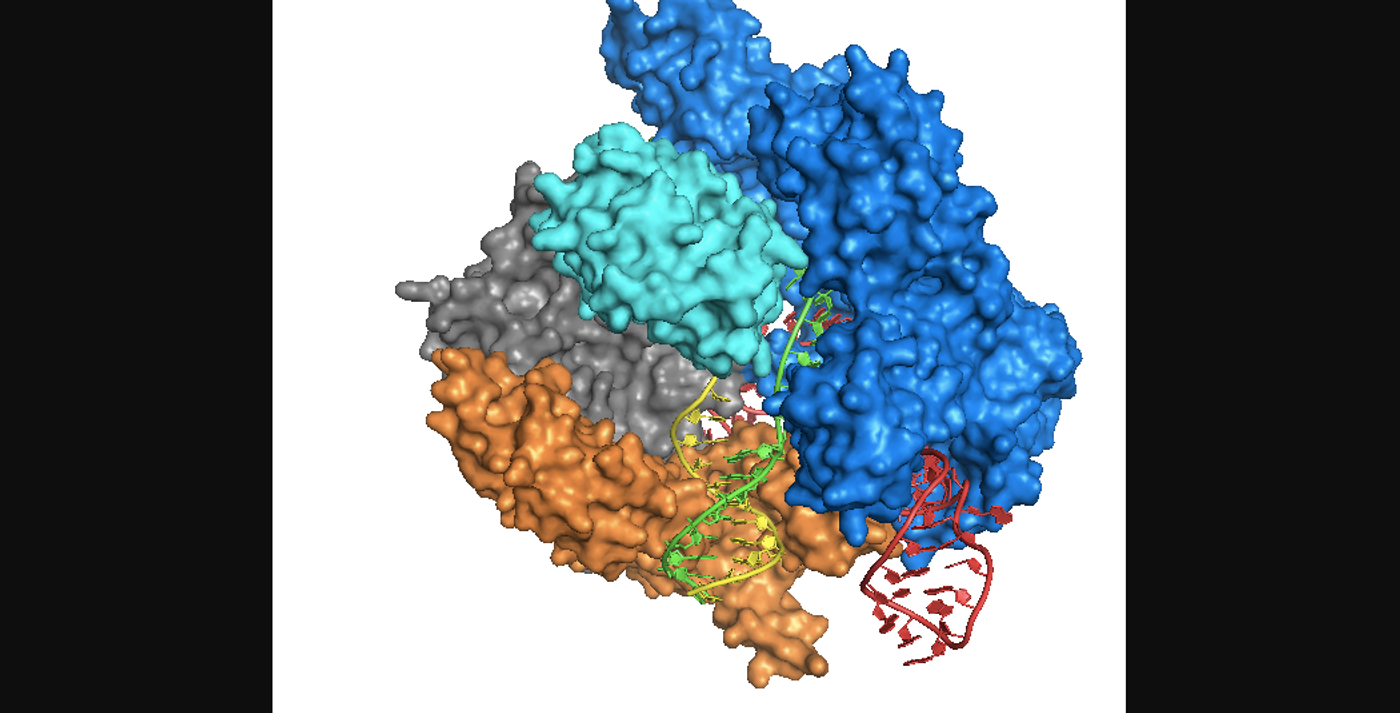
The new system was reported in Nature Biomedical Engineering by researchers led by Berkeley bioengineering professors Niren Murthy and Irina Conboy. It uses gold nanoparticles to deliver the Cas9 enzyme along with a guide RNA and donor DNA into live cells of an organism. "CRISPR-Gold is the first example of a delivery vehicle that can deliver all of the CRISPR components needed to correct gene mutations, without the use of viruses," Murthy said.
The gold nanoparticles coat donor DNA and also binds to Cas9. The donor DNA gets added to the genome as part of a DNA repair system used by the cell, homology-directed repair. When CRISPR-Gold was tested in mice, cells imported the editing components after recognizing a marker on the CRISPR-Gold. After getting into the cell, it breaks apart and releases the system.
One CRISPR-Gold injection into mouse tissue repaired 5.4 percent of dystrophin gene mutations, which cause the disease, in a Duchenne muscular dystrophy model. That was a rate roughly 18 times higher than in mice that were injected with Cas9 and donor DNA alone; that group showed a dismal 0.3 percent correction rate.
"These experiments suggest that it will be possible to develop non-viral CRISPR therapeutics that can safely correct gene mutations, via the process of homology-directed repair, by simply developing nanoparticles that can simultaneously encapsulate all of the CRISPR components," Murthy noted.
An assessment of the safety of this system showed good results. It has less toxicity, fewer side effects, and had off-target effects that were comparable to DNA errors in a cell that has not been exposed to CRISPR. It also did not affect the immune system.
"CRISPR-Gold and, more broadly, CRISPR-nanoparticles open a new way for safer, accurately controlled delivery of gene-editing tools," Conboy said. "Ultimately, these techniques could be developed into a new medicine for Duchenne muscular dystrophy and a number of other genetic diseases."
More work will be necessary, including a clinical trial, before this tool can be used in humans. The researchers are interested in targeting adult stem cells with CRISPR, and several have collaborated on a start-up venture.
"Genetic diseases cause devastating levels of mortality and morbidity, and new strategies for treating them are greatly needed," said Murthy. "CRISPR-Gold was able to correct disease-causing gene mutations in vivo, via the non-viral delivery of Cas9 protein, guide RNA and donor DNA, and therefore has the potential to develop into a therapeutic for treating genetic diseases."
Check out the video above to learn more about how CRISPR is revolutionizing mouse models of disease in myriad ways.
Sources: ScienceDaily Via University of California Berkeley, Nature Biomedical Engineering