Editing Genes in the Brain with CRISPR/Cas9
Relatively recent advancements in gene editing tools have entirely revolutionized biomedical science. There are, of course, limitations to the technology. One major caveat is that cells have to be dividing for it to work. That immediately excludes most cells in the brain, because those neurons don’t often divide. Researchers are aiming to change that, however. New work has tweaked CRISPR/Cas9 gene editing technology so it can be used in non-dividing neurons. The study is summarized in the following video.
Research Fellow Jun Nishiyama, M.D., Ph.D., Research Scientist, Takayasu Mikuni, M.D., Ph.D., and Scientific Director, Ryohei Yasuda, Ph.D. of the Max Planck Florida Institute for Neuroscience (MPFI) have created a new way to precisely edit the genome of mature neurons for the first time. This is a significant advancement that is expected to opening up new avenues for neuroscience.
CRISPR/Cas9 takes advantage of mechanisms used by bacterial immune systems and targets a sequence of DNA so for a cut. When that cut is repaired, cellular machinery is usually used in a process called non-homologous end joining (NHEJ) repair. NHEJ tends to introduce some small errors. Another repair process, homology-directed repair (HDR), is much more precise and can change the genome in very specific ways.
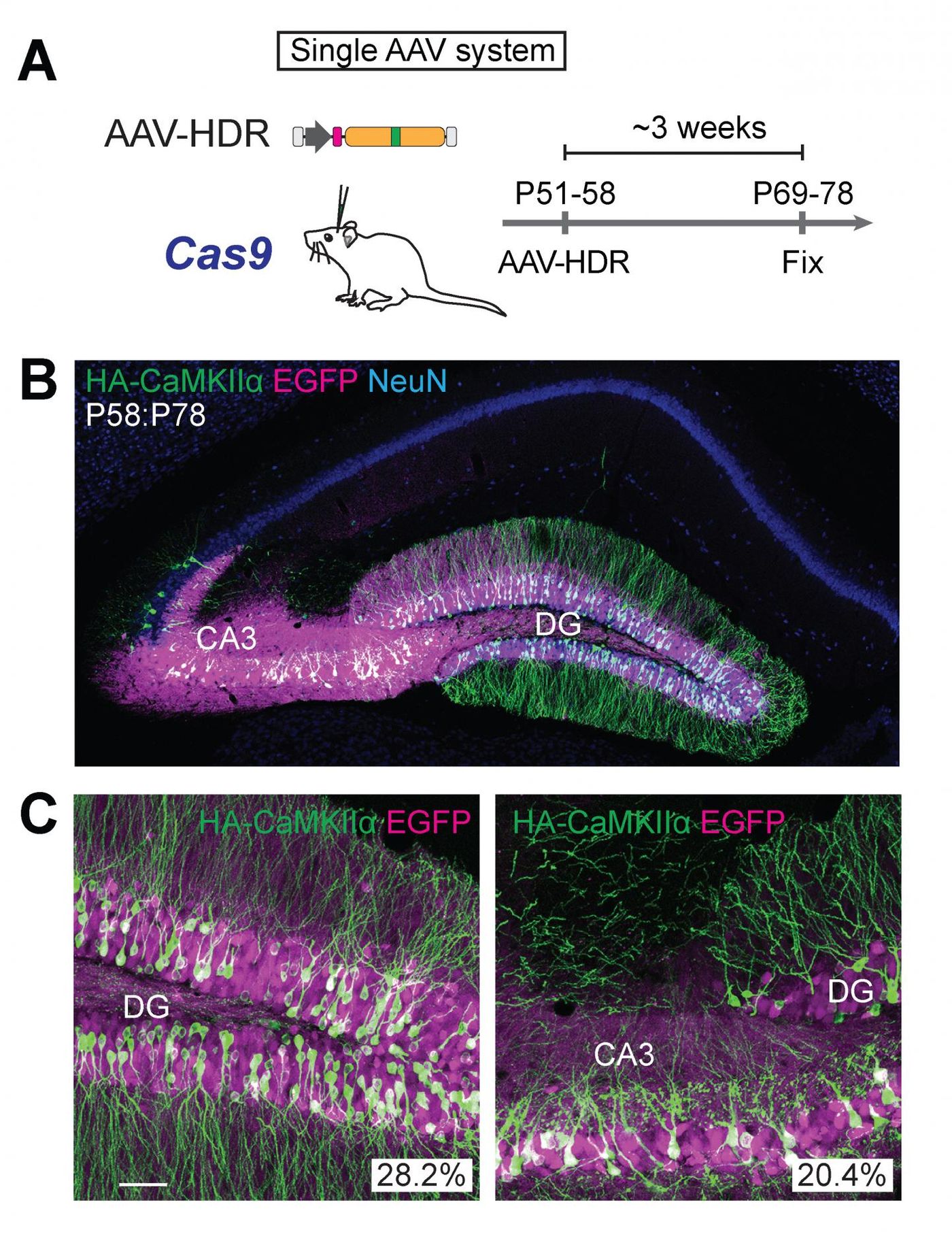
HDR was thought to only apply to cells that can still undergo division, and cells in the brain are thought of as non-dividing, or post-mitotic. As such, it was assumed that the precise gene edits that HDR and CRISPR/Cas9 make possible could not be used on neurons in the brain. However, the researchers added a virus to the gene-editing cocktail. Now, post-mitotic neurons in the brain can be targeted by HDR in a technology named vSLENDR, for viral-mediated single-cell labeling of endogenous proteins by CRISPR/Cas9-mediated homology-directed repair.
A commonly used virus that is nontoxic and elicits little response from the immune system, adeno-associated virus (AAV) is often used by scientists for the efficient delivery of many kinds of genes. The virus can be used as an efficient repair template for HDR. Transgenic mice expressing Cas9 showed HDR in post-mitotic neurons of the brain after being exposed to the editing machinery and the AAV.
The researchers also tested their work in mice that did not express Cas9, but instead model Alzheimer’s disease. The therapeutic potential of the work was highlighted; the vSLENDR technique worked in these aged mice.
The new tool has an amazing range of potential application in both the clinic and in research science. It may accelerate research as well as aid in the development of new therapies for neurological diseases.
Sources: Max Planck Florida, Neuron