Novel Chemistry That Allows Faster and Easier Synthesis of New PET Scan Probes
For positron emission tomography (PET) scans, patients are injected with a radioactive probe that emits positrons (a positron is the antimatter of an electron) and tracks specific tissues or metabolic activities. The locale, size, and intensity of the scanner-captured signals would enable radiologists to perform proper diagnostics of diseases.
A group of radiochemists at the UNC Lineberger Comprehensive Cancer Center has developed a new fluorination mechanism. Using this new technique, they can radiolabel a large variety of small molecules and prepare PET scan tracers that were deemed too challenging and not feasible.
As an element, fluorine has the highest electronegativity, meaning the atom has the highest potential to capture an electron to become isoelectronic. With a half-life about 110 minutes, fluorine-18 (F-18) is a favorable choice for making PET diagnositics.
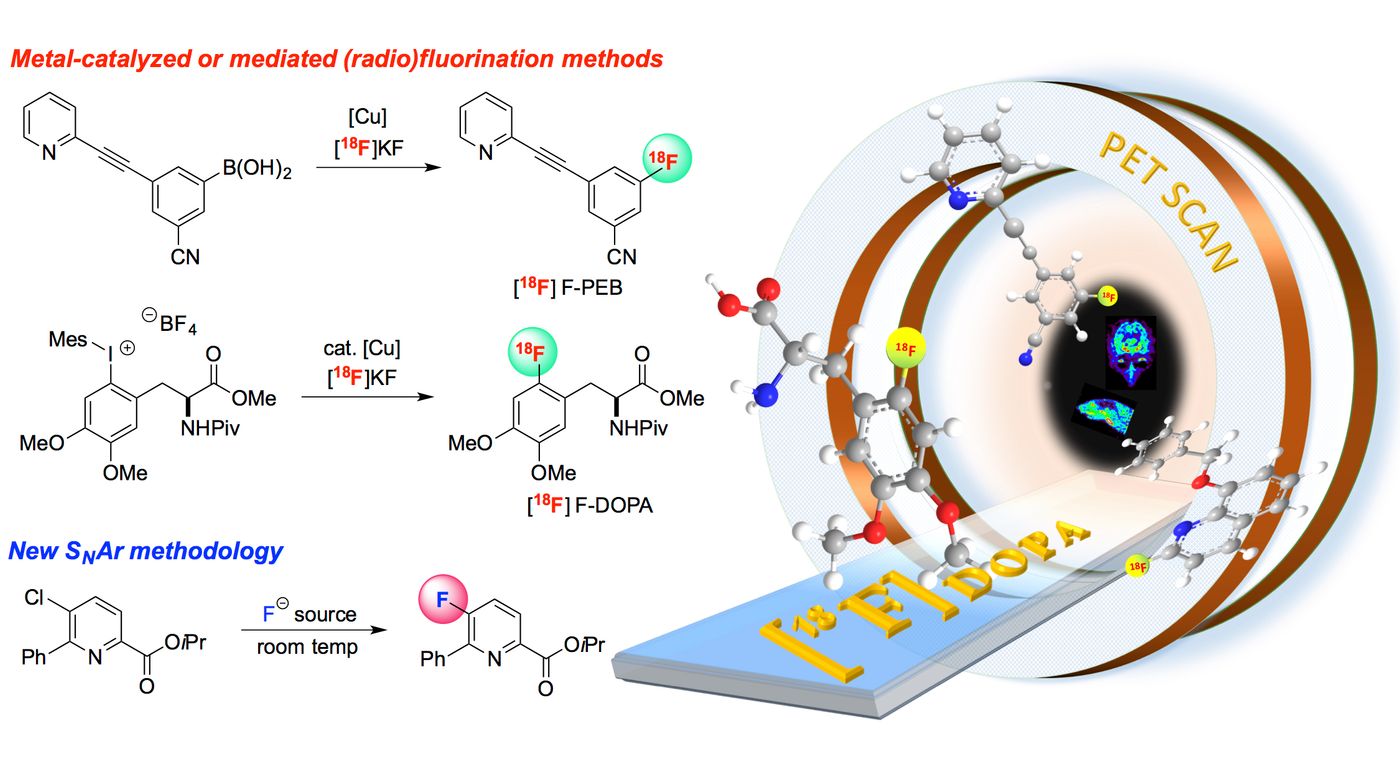
There are two ways to add a fluorine atom to a molecule. One route is through electrophilic substitution, which involves decreasing the electron density on fluorine (F2 is often used as a fluorinating agent in such reaction). But electrophilic fluorination methods are often inefficient and have a low yield, which in turn resulted in a low molar activity. Imagine a large pool of molecules are placed in a reaction, but only a handful of them get tagged with fluorine, thereby separating them would be like finding a needle mixed in a haystack.
On the other hand, the nucleophilic fluorination route is much more favorable because of the higher molar ratio in products. But the process cannot directly fluorinate molecules by substituting the C-H, for it requires specific precursors such as halogenated aromatic hydrocarbons, aryl boronic acids, or aryl azides. Synthesizing these precursors demand extra time and cost. In many current methods for making F-18-based PET probes, transition metals such as nickel and palladium are commonly used as catalysts. However, they are incompatible with human consumption, unless an extra separation step is added to remove the metals before administration.
The new method brought forward by the UNC researchers is quite different in the sense that it does not require pre-made precursors, nor does it need metal catalysts. They incorporated an acridinium (a chemiluminescent) and a 3W blue light laser at 450 nm, as photo-redox catalytic components to directly substitutes aryl carbon-hydrogen bonds with F-18 anion. The technique was experimented to attach the radioactive fluorine to a variety of molecules with aromatic rings, most of which resulted in a molar activity that is suitable for the synthesis of PET probes.
How does a PET scan work? (Imperial College London)
Using this novel mechanism, the team is hoping to rapidly develop a pool of PET tracers directly from bioactive molecules. What's more, their radio-fluorination method can also enable drug discovery by generating radio-fluorinated analogs of drug candidates and evaluating their distribution profile through animal PET imaging.
This ground-breaking chemistry research was recently published in the journal Science.
Source: MedicalXpress