Using Bacteria to Reduce Carbon Dioxide Emissions
The bacteria Clostridium thermocellum is an attractive candidate for potential use in biofuel production due to its cellulolytic abilities. This capability to digest plant material into biofuels and chemicals without the separate addition of other enzymes could increase ease and efficiency of the reactions. However, the mechanism by which the bacteria are able to metabolize cellulose and consume carbon dioxide, or CO2, has not been discovered. By consuming the standard CO2 byproduct from the initial reaction, the bacteria open up an entirely new route of greener biofuel production and potential reduction of greenhouse gasses.
Lisa Warner, an assistant research professor in the Biomolecular Research Center at Boise State University and colleagues affiliated with the U.S. Department of Energy’s National Renewable Energy Laboratory were able to identify the uncharacterized metabolic pathway used by these bacteria. While scientists have known that some bacteria are capable of CO2 uptake, they have not been able to figure out how. This study enables researchers to increase understanding of this process and make use of the newly identified enzymes involved in the process to develop production procedures that result in a decrease in toxic emissions.
“Now that we’ve shown we have a novel pathway we can take advantage of the enzymes that convert CO2 to something else and create a cocktail of molecules with the least amount of noxious byproducts,” Warner said.
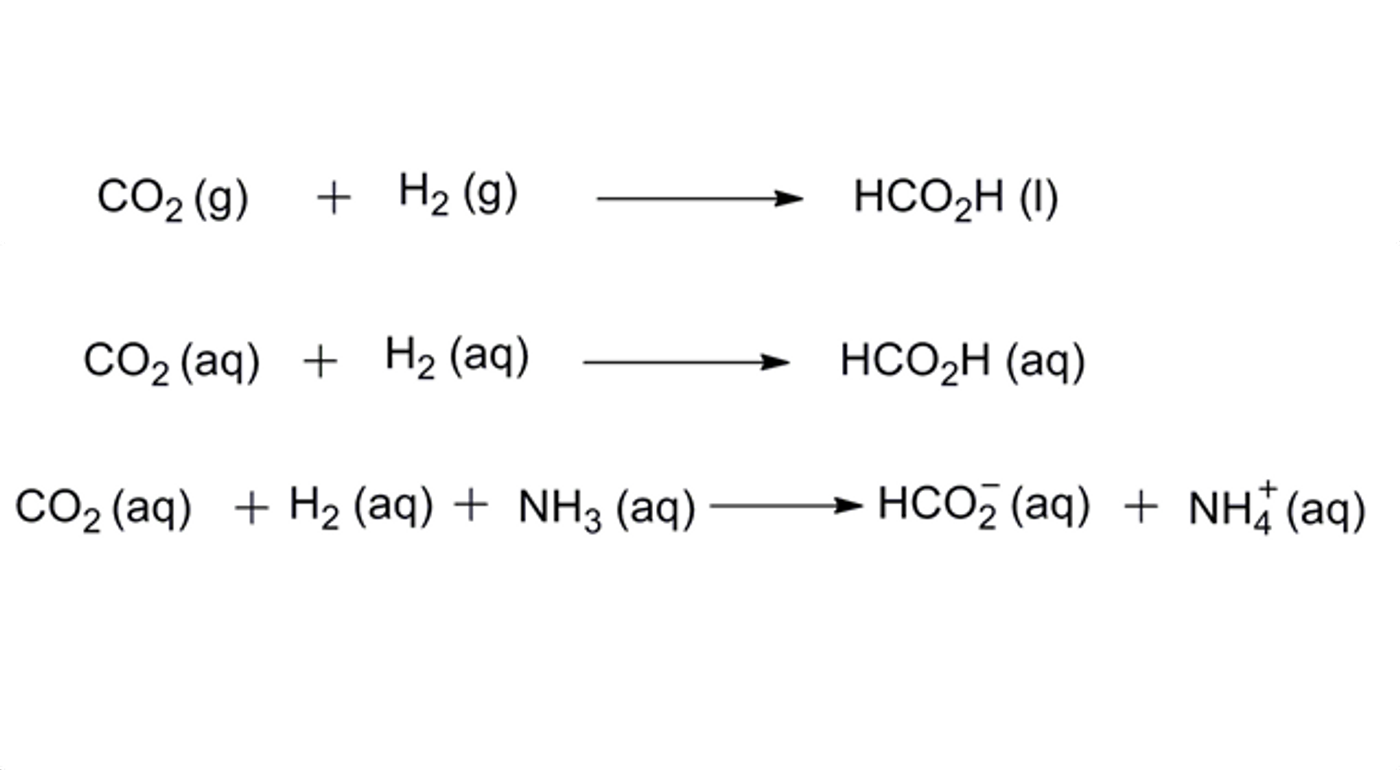
Clostridium thermocellum is one of the most efficient converters of plant material into hydrogen and hydrocarbon biofuels. CO2 is a standard byproduct of the reaction, causing concern about contributing to the already high CO2 levels in the atmosphere. The researchers discovered that these bacteria, however, are able to reclaim some of the CO2 byproduct and convert that into formate, or HCO2, under certain conditions.
The obstacle is that formate and CO2 look almost identical in analysis with a mass spectrometer, so discriminating between the two within the pathway has proven difficult. Warner and her collaborators were able to distinguish the two molecules using nuclear magnetic resonance spectroscopy.
Since Clostridium thermocellum normally lacks the ability to convert CO2 into formate, the team surmised that addition of 13C-bicarbonate used to promote bacterial growth through additional carbon metabolism might stimulate CO2 conversion. When the bacteria were fed 13C-bicarbonate, a dissolved form of CO2, a novel pathway was initiated that was capable of catalyzing the reduction of CO2 to formate. Combining genomic and experimental data, the team demonstrated that the conversion of CO2 to formate can serve as CO2 fixation in a biofuel production reaction and channels the CO2 into a unique carbon metabolic pathway that internalizes CO2 via two biochemical reactions.
The use of carbon isotopes allowed the group to track CO2 upon uptake and determine how it incorporates into new products. The findings highlight the metabolic versatility of Clostridium thermocellum and the capability to not only play a primary role in biofuel production but also in CO2 fixation as well. The study was published in the journal Proceedings of the National Academy of Sciences of the United States of America.
Sources: Biomass Magazine, PNAS, Microbiology Online