Proton Therapy Can Become More Accessible to Cancer Patients after Some Tweaking
If there are only two things to be said about proton therapy, the first is that it is superior in treatment quality for cancer patients, and the second is that the technology bears a considerable cost which in return falls on the shoulders of patients and healthcare insurers. According to the two experts, Thomas R. Bortfeld and Jay S. Loeffler medical physicists from Massachusetts General Hospital, some aspects can be improved so that more patients with localized tumor can get access to proton therapy.
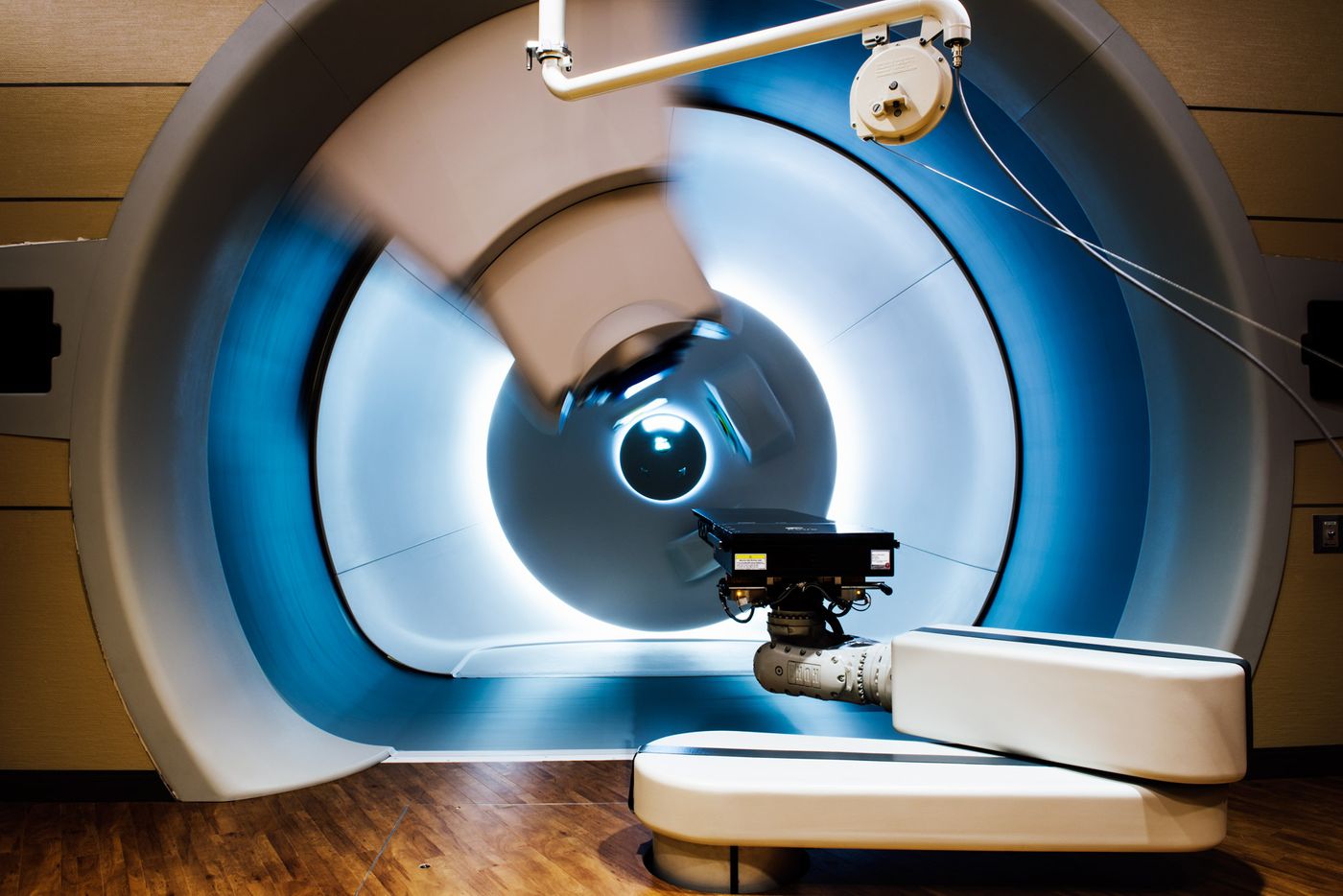
Treatment room of the Texas Center for Proton Therapy at Las Colinas. Credit: D Magazine
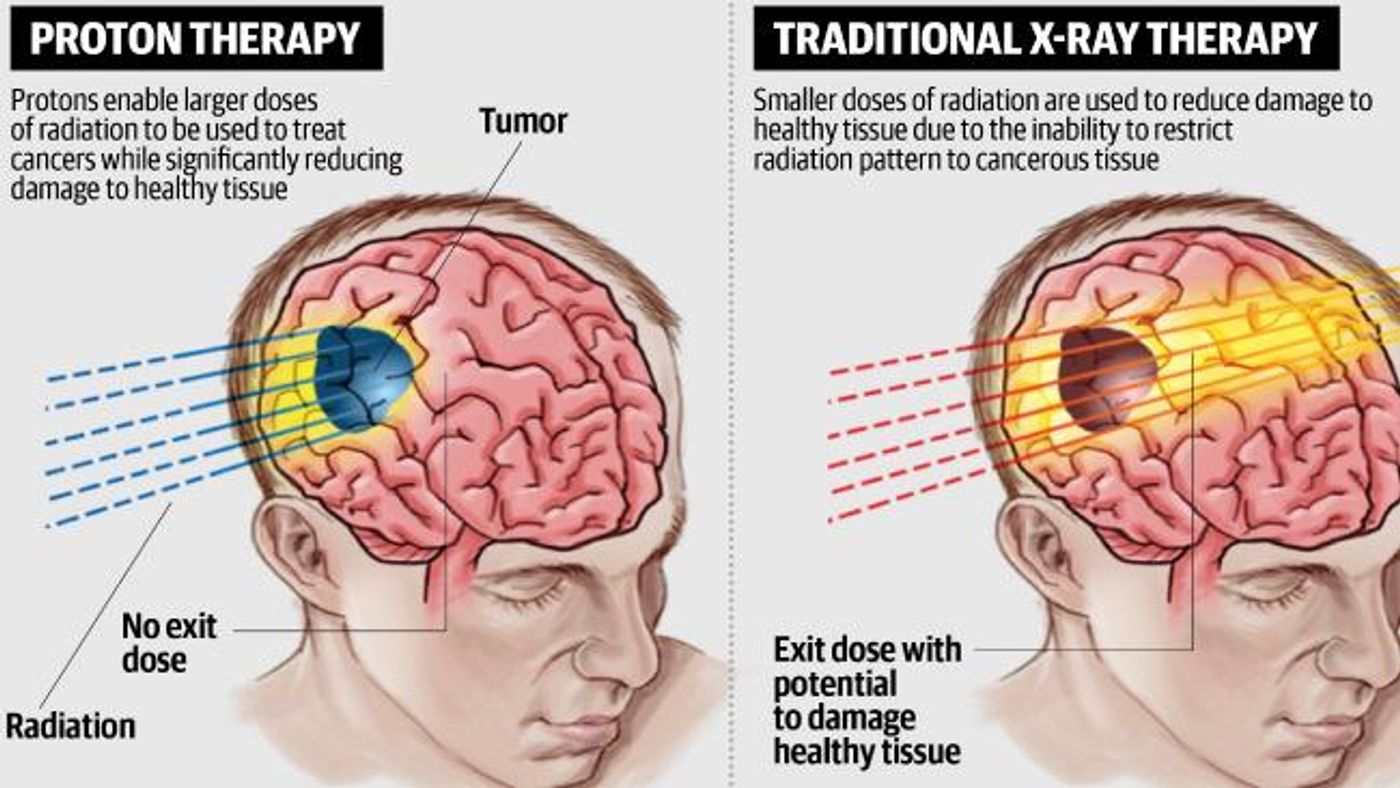
Proton therapy uses a beam of protons to irradiate cancerous tissues. Like other types of radiation therapy, it works by breaking up the DNA double strands in the tumor cells. The damage is either direct or indirect ionization of the atoms which make up the DNA chain.
In photon therapy such as X-rays or gamma rays, most of the radiation effect is through free radicals, a result of the ionization of water. Its effectiveness is hindered when facing cells of solid tumors that are deficient in oxygen. Low oxygen state is bad for forming free radicals.
Charged particles such as protons ions can cause direct damage to the cancer cells, and its tumor-killing effect is independent of tumor oxygen. These particles act mostly via direct energy transfer which results in the breaking of DNA double-strand.
Also due to their relatively large mass, protons have little lateral side scatter in the tissue, meaning that the beam stays focused on the tumor shape and delivers small dose side-effects to surrounding tissue.
Difference between traditional radiation therapy and proton therapy. Credit: DailyTelegraph
In the commentary published in Nature, Bortfeld and Loeffler argued that by resolving the following three issues: reducing the size of particle accelerators, sharpening proton beams and broadening health-care coverage, more cancer patients can benefit from the advanced treatment.
Proton beams are generated by particle accelerators like cyclotrons or synchrotrons. In the past decades, both the weight and size of accelerators has gone down a lot, and the development of more compact accelerator systems including superconducting synchrocyclotrons, ultra-compact synchrotrons, dielectric wall accelerators, and linear particle accelerators are underway. Currently, the smallest therapeutic accelerator is approximately the size of a king-sized bed.
However, the total footprint of the combined pieces of equipments still take up a couple of hundred square meters or above 2,100 square feet. Most cancer hospitals had neither the cash nor the space to house what are required for proton therapy. Both the equipment and the price tag need to shrink further if proton therapy is to outcompete photon-based radiation therapy.
Sharpening the proton beam can improve the precision of therapy. While high energy photons go right through the tissue, fast protons that enter the patient can see a quick slow down due to their interaction with body tissues, resulting to the point past which no more effect can be seen. Most of the beam's energy is deposited at the point, like the tip of a scalpel.
The speed of the proton dictates how deep the beam can reach into the human body. For example, protons with energies of around 50 MeV (mega electronvolt; 1 eV equals the amount of energy gained or lost by a single electron moving across an electric potential difference of one volt) can penetrate to a depth of a few centimeters; whereas with over 200 MeV, the proton can reach 30 cm. Further refining the focus of the proton beam and lower the overspill of radiation would even make it an ideal treatment for all oncology patients who need radiation therapy.
Coverage-wise, there are a lot more needed to accomplish to push proton therapy to the forefront. Currently, in the US, some hospitals focus on common and easy-to-treat tumors such as prostate cancer so that they can recover the investment on proton centers. Those who can afford the treatment often pay out of their pockets, since insurers are reluctant to cover such treatment. So the most common cancer treated with protons is one in which it makes the least clinical difference.
Another accessibility issue is that patients must be referred to a handful of therapy centers. Domestic or even international traveling expenses and the local physicians’ fear of losing revenue make few patients benefit from the technology.
By tackling all three bottlenecks, Bortfeld and Loeffler hoped that proton therapy could one day become the treatment of choice for all eligible cancer patients.
Cyclotron and Proton Therapy Animation. Credit: Scripps Proton Therapy Center
Source: Nature