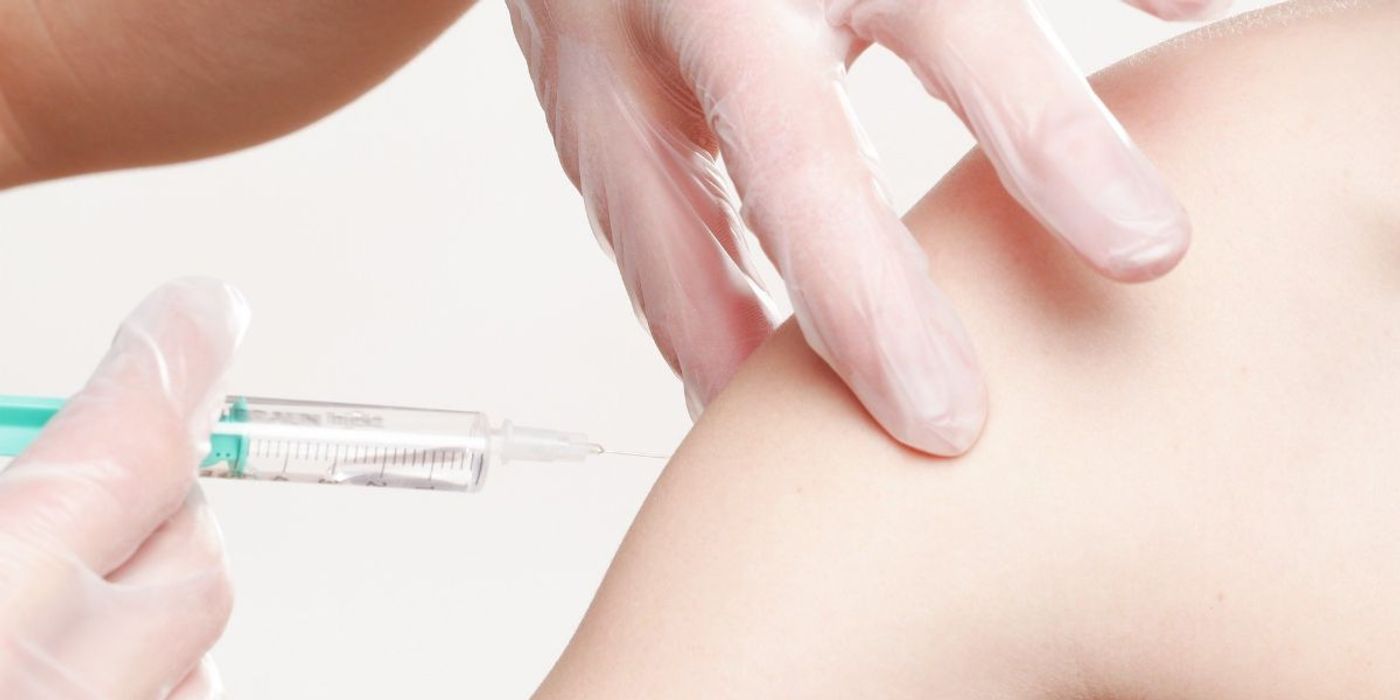
COVID-19 Vaccine by Pfizer More than 90% Effective
A preliminary analysis of Pfizer and BioNTech's COVID-19 vaccine shows that it can prevent over 90% of people from contracting COVID-19.
For the study, researchers tested the vaccine on over 43,000 people across six countries: the US, Germany, Brazil, Argentina, South Africa, and Turkey. During the Phase 3 trial, volunteers either received the COVID-19 vaccine, administered in two shots around three weeks apart, or a placebo alternative such as a meningitis vaccine. Neither doctors nor patients knew which they had.
So far, the results look positive. From an interim analysis of the first confirmed 94 cases of COVID-19 among those involved in the research, scientists found that fewer than 10% of infections- or around eight people- had received the vaccine. This means that over 90% of the cases were people who had received the placebo.
Pfizer has said that the vaccine had an efficacy rate of more than 90% at seven days after the second dose, meaning that protection is achieved within 28 days of beginning the vaccination process. While the companies behind the vaccine aim to have it approved by the end of the month, they aim to be able to assess 164 confirmed cases of COVID-19 infection among trial participants before final results are published.
The vaccine itself is based on a new approach involving messenger RNA (mRNA). Unlike other vaccines that directly use the virus, this vaccine uses the virus's mRNA to cause cells in the body to produce proteins that resemble the virus. This then triggers an immune response that recognizes and attacks these proteins, which are theoretically summoned again if the virus enters the body.
While the vaccine is deemed to be effective, the CEO of Pfizer, Dr Albert Bourla, said that it is still unknown how long protection from the vaccine will last. He added that it is likely that people will need periodical vaccinations to ensure immunity. Regardless, Bourla has said that Pfizer expects to produce 50 million vaccine doses by the end of this year and 1.3 billion next year.
Sources: BBC, CNN, The Guardian