Spun Threads and Computer Models Determine the Best Dose for Pancreatic Cancer Drug
Image Credit Jenner et al 2023 via Creative Commons license CC BY 4.0, and Francois Bonvin’s A Woman Spinning Flax via Creative Commons license CC0 1.0, alterations made to combine images by Amielle Moreno
Pancreatic cancer is projected to surpass breast, prostate, and colorectal cancer to become the second leading cause of cancer-related deaths in the United States by 2030. If untreated, this diagnosis usually indicates 3-to-5-month survival.
It’s not as though chemotherapeutic drugs such as gemcitabine aren’t effective; pancreatic ductal adenocarcinoma (PDAC) tumors are just incredibly dense. Their fibrous connective tissues and collapsed blood vessels lead to poor uptake of chemotherapeutic drugs.
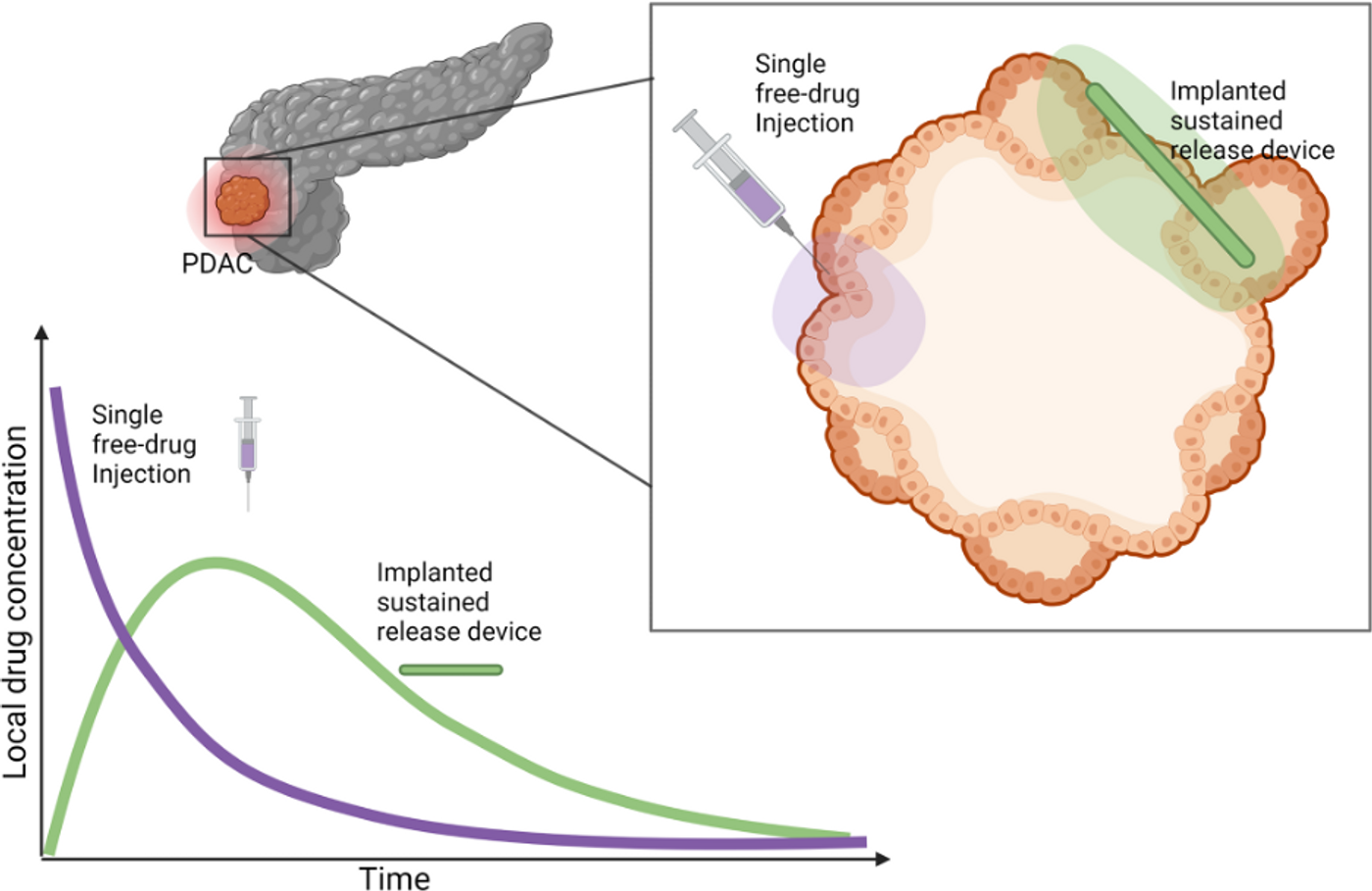
Researchers at Queensland University of Technology are exploring a potential solution that involves implanting drug treatments directly at the site of the tumor. In 2017, the laboratory of Kara L. Vine published a study describing the development of chemotherapeutic polymer fibers. The chemotherapeutic cancer drug gemcitabine was encapsulated within these 50-120 micrometer thick stands.
Treating human pancreatic cancer cell lines with drug-loaded fibers decreased the cell growth rate. Whereas intravenous delivery of chemotherapy is known to place stress on non-cancer cells, this treatment option offers to treat tumors locally. Threads implanted into the tumor decreasing the rate of toxicity associated with chemotherapy.
Now, from the same laboratory, Adrianne L. Jenner and collogues published computational models of both PDAC growth and the impact of their drug delivery system in PLoS Computational Biology.
Using in vitro and in vivo data on PDAC growth, the researchers designed a valuable computational model of pancreatic tumor cell growth and death. They paired this with a predictive model of their spun fiber drug delivery system.
The combination of computational models can determine the optimal drug release profiles and implantation protocols that have yet to be tested experimentally.
This new in silico experimental approach offers predictive insight into the effect of implant treatments on these tough-to-beat tumors and places future researchers and clinicians at an advantageous starting point.
Image Credit Jenner et al 2023, via Creative Commons license CC BY 4.0, no alterations made.
In the future, the model can be expanded to consider polymers with multiple drug loads to help clinicians deliver the perfect one-two punch to the terrifying tissue. It also has applications for other types of cancers and drugs.
Sources: PLoS Computational Biology, Pancreatotomy, Cancer Research