Recombinant Antibody Production in Drug Discovery
Therapeutic antibodies are one of the best-selling drug classes in the pharmaceutical market, and the advent of antibody engineering and recombinant production dramatically improved the arsenal of therapeutics against acute and chronic diseases.
Introduction
Antibody engineering and production are essential tools in drug discovery, providing powerful and specific therapeutics and diagnostic/prognostic materials for various diseases. Antibodies are proteins produced by the immune system in response to foreign invaders, such as bacteria or viruses. They can bind specifically to these invaders, neutralizing and preventing them from causing harm. Over the last few decades, scientists have developed methods for engineering and producing antibodies that can be used as drugs. This article will provide an overview of the process of antibody engineering and production, as well as their applications in drug discovery.
History
The history of antibody engineering and production in drug discovery can be traced back to the 1970s and 1980s1, when scientists first began to explore the potential of using antibodies as therapeutics. The first generation antibodies were derived from natural sources, such as animal blood, and were used primarily as diagnostic reagents.
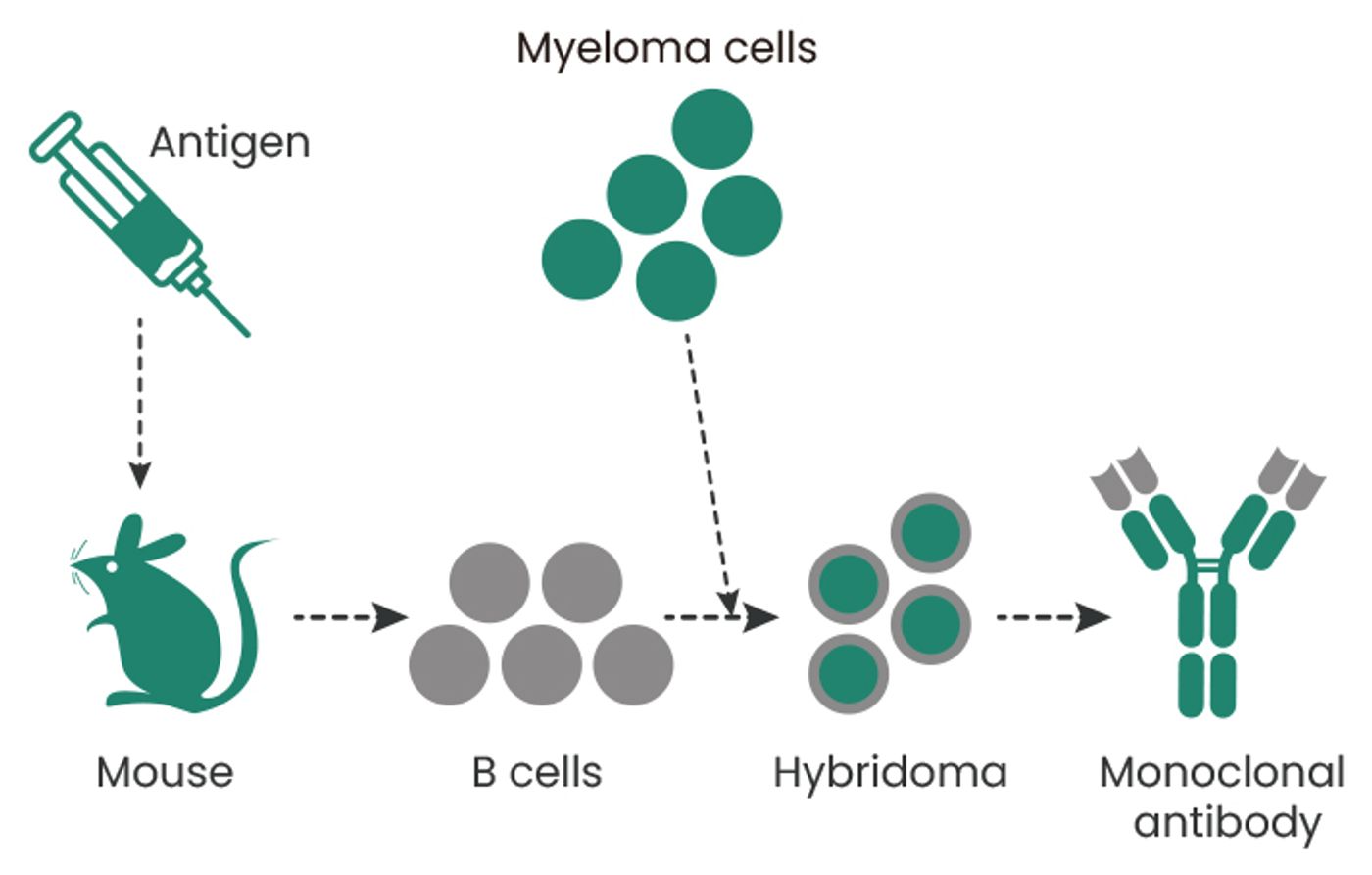
One of the key early milestones in the history of antibody engineering was the development of hybridoma technology in 1975 by César Milstein and Georges Köhler. This allowed for the production of monoclonal antibodies (mAbs) by fusing a B cell, which produces a single antibody type, with a myeloma cell that can divide indefinitely (Figure 1). This made it possible to produce large quantities of specific antibodies. However, the production of early mAbs was limited by the availability of suitable myeloma cell lines (usually mouse or rat) and the fact that hybridomas can be low yielding or genetically unstable2.
Figure 1. Hybridoma technology.
In the 1980s, scientists began to develop methods to produce recombinant antibodies, which are made using genetic engineering techniques. These methods allowed scientists to produce large quantities of pure and consistent antibodies in a controlled environment. This was a significant step forward, as it made it possible to manufacture antibodies on a large scale for use in preclinical and clinical trials.
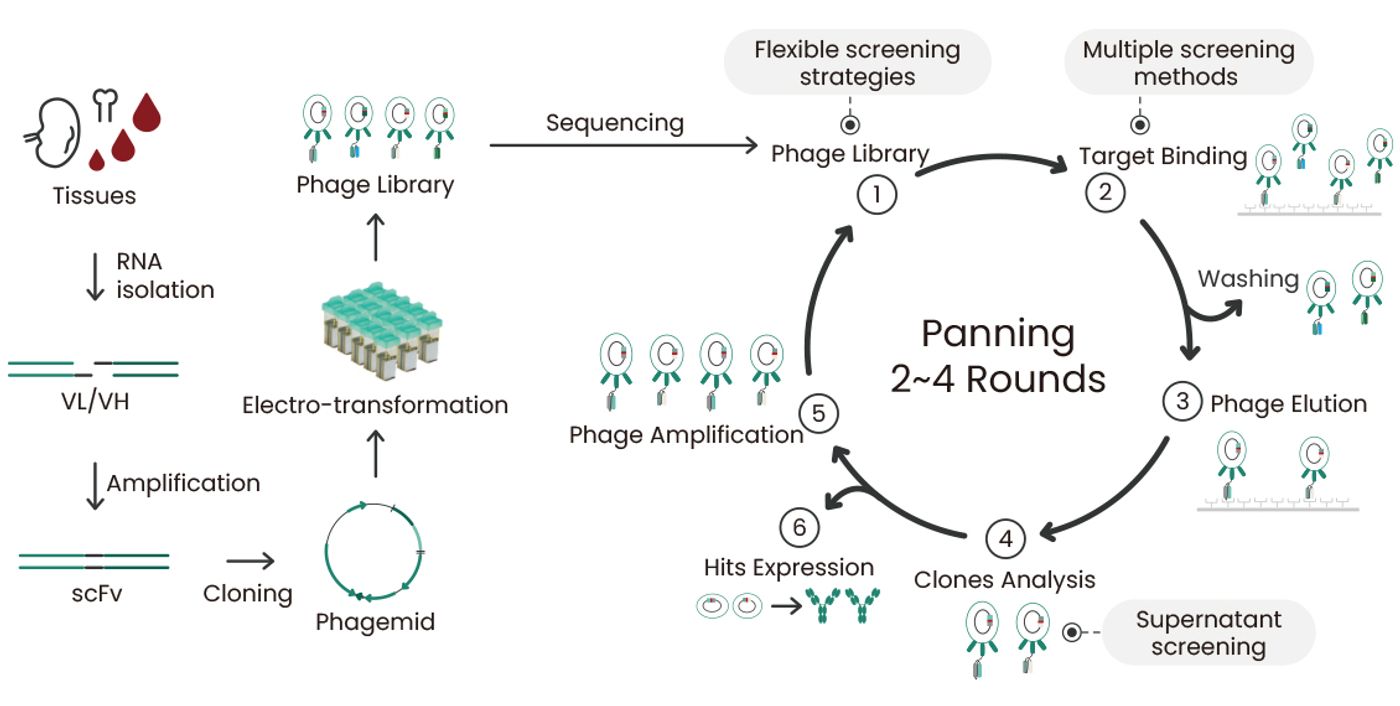
Then, about one decade later, the development of display technologies, such as phage display, yeast display, and ribosome display, made it possible to screen large libraries of antibodies for specific binding properties. This quickened the process of identifying antibodies that bind to specific antigens. This also made it possible to optimize the structure of antibodies to improve their binding properties using mutant libraries. Such display technologies, especially phage display, are a huge driving force for discovering numerous antibodies that can be further engineered, optimized, and recombinantly produced3. Phage display antibody library technology involves multiple steps, including the construction of phage display antibody libraries, panning, mAb identification, and recombinant expression of positive clones (Figure 2). Sino Biological is specialized in recombinant protein production and antibody development. Based on its expertise in the construction and screening of phage display antibody libraries, Sino Biological provides efficient antibody discovery services.
Figure 2. Technical route of phage display library construction and screening.
The first licensed mAb drug is muromonab-CD3 (1986), a hybridoma derived antibody that was used for immunosuppression during organ transplantation. Rituximab (Rituxan), approved by the FDA in 1997, targeted CD20 protein in B-cell non-Hodgkin’s lymphoma. It marked a significant milestone in the field of antibody engineering and production as it was the first antibody drug made with recombinant technology. Rituximab was also the first mAb approved for cancer treatment, followed by a number of other mAbs in the following years. Since then, approximately 170 antibodies and antibody-based therapeutics (including antibody-drug conjugates) have been approved or are under review by authorities worldwide4,5.
Antibody optimization
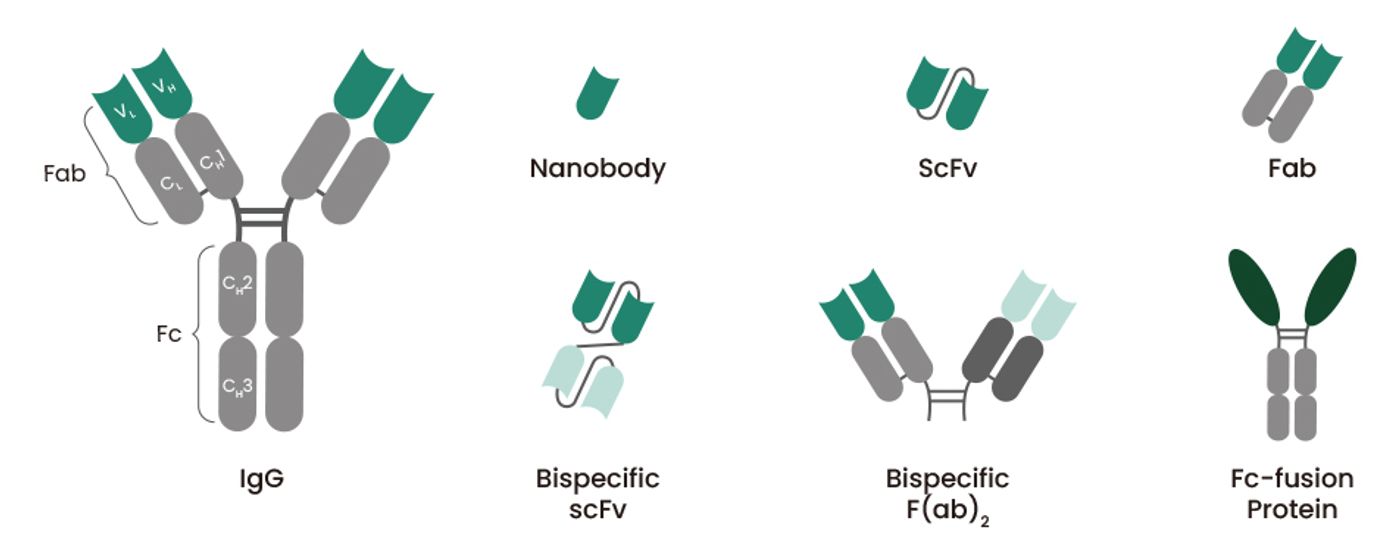
Besides the ability to produce large quantities of antibodies with a controlled quality, benefits of recombinant antibody production include the abilities to generate different antibody structures and derivatives (Figure 3), such as bispecific antibodies (bsAbs) and Fc-fusion proteins, and to optimize antibody structure, which can be done by introducing mutations into antibody genes, in order to improve the clinical effectiveness of antibodies.
Figure 3. Examples of recombinant antibody formats.
BsAbs, designed to bind simultaneously two different antigens or epitopes, have a wide range of applications, such as redirecting specific immune effector cells to tumor cells and blocking two different pathways. Fc-fusion proteins, composed of Fc region of IgG antibody and a desired linked protein, such as receptor extracellular domains, enzymes, cytokines, and active peptides, have additional beneficial biological and pharmacological properties. The primary reason for fusing a biologically active protein of interest with Fc is that the Fc domain contributes to the plasma half-life extension. Sino Biological has extensive experience in producing recombinant antibodies in diverse formats and provides a variety of service packages to meet different research and drug discovery needs.
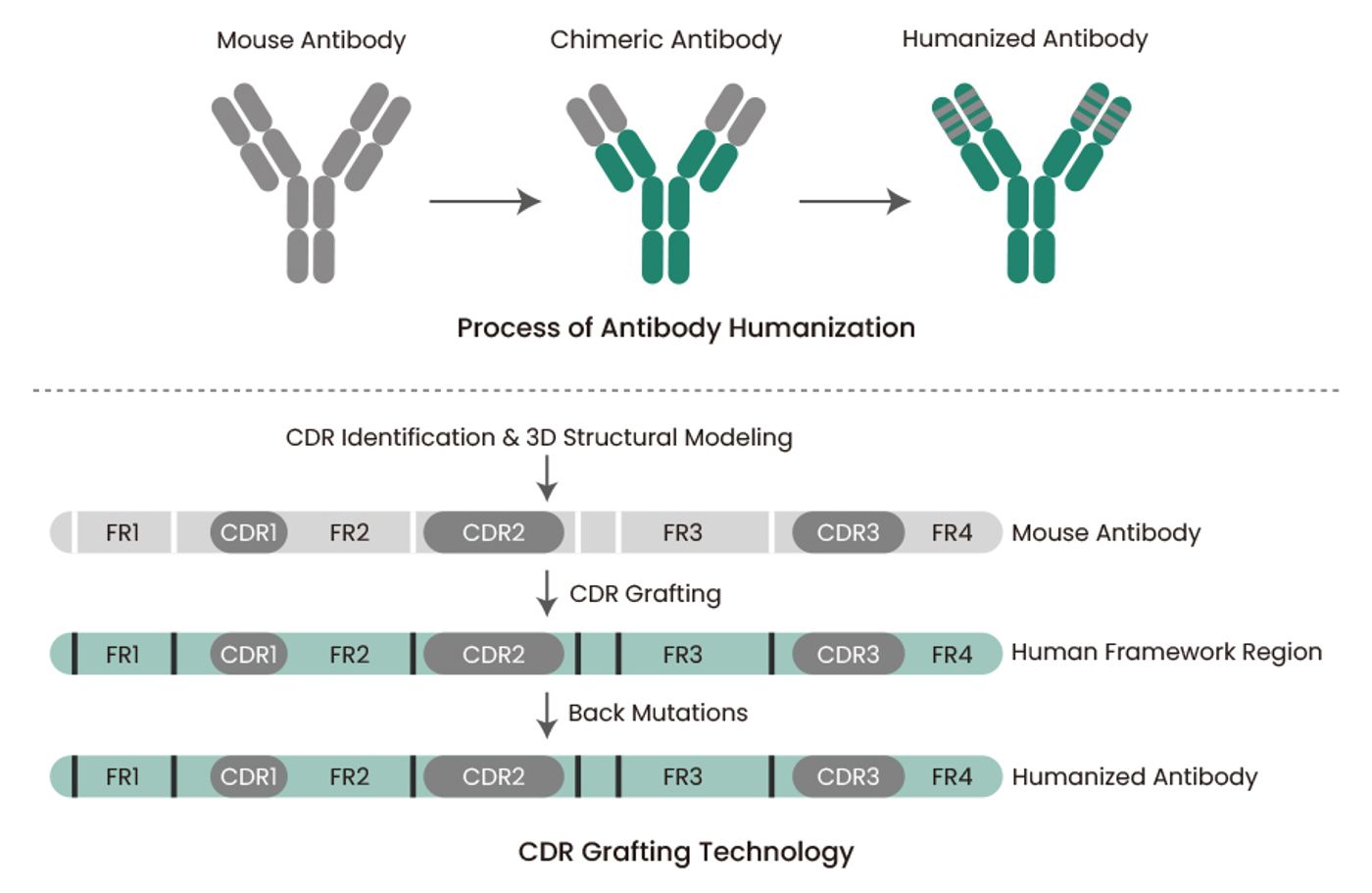
Antibody humanization is one of the key antibody optimization technologies. Humanized antibodies represent a major type of antibody drugs. The clinical application of mouse mAbs is limited by the human anti-mouse antibody (HAMA) response, which not only neutralizes these therapeutic antibodies but also leads to allergic response in patients. Humanization of mouse mAbs by genetic engineering can minimize their heterologous nature as well as maintain their specificity and affinity, which can improve the safety and therapeutic efficacy of mAbs in clinical applications. Sino Biological provides high-quality mAb humanization services using complementarity-determining region (CDR) grafting technology and computer-aided molecular modeling (Figure 4).
Figure 4. Process of antibody humanization and CDR grafting technology.
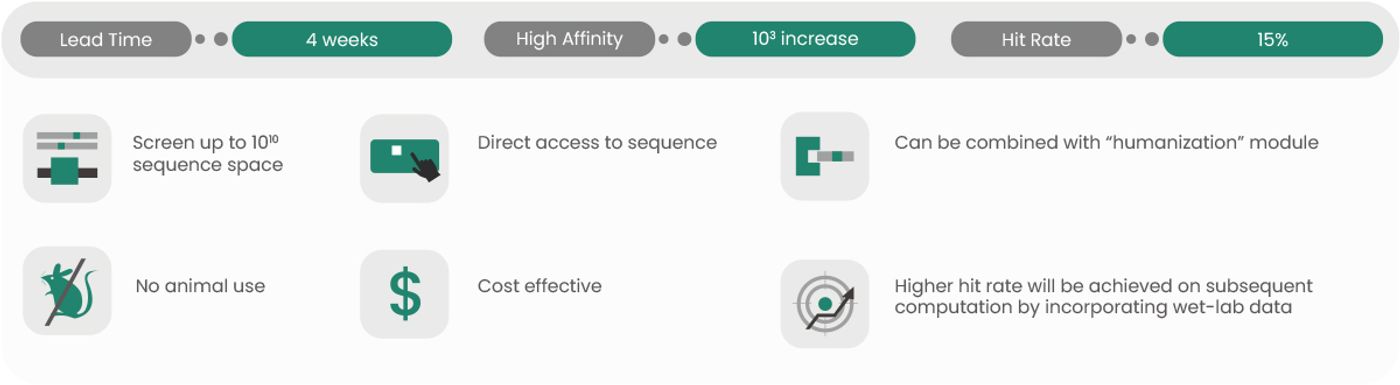
More optimization methods are evolving as the field of antibody engineering and production continues to expand. For instance, scientists can optimize the way an antibody binds to its antigen using techniques such as artificial intelligence (AI)-driven affinity maturation performed in silico instead of relying on in vivo affinity maturation that requires animal use. Sino Biological provides AI-powered affinity maturation service, which is a computational and experimental service aimed at improving the affinity and specificity of mAbs using machine learning algorithms to predict the effect of mutations on antibody-antigen binding and then validating the predictions in wet lab (Figure 5). This service is useful for researchers who are developing mAbs for therapeutic or diagnostic applications, making their candidate antibodies more effective.
Production method
Once the desired antibody sequences are obtained, they must be produced in large quantities for use in drug development. This is typically done by growing large numbers of cells, such as Human Embryonic Kidney 293 (HEK293) cells or Chinese hamster ovary (CHO) cells, which have been genetically modified to produce the desired antibodies. CHO and HEK293 cells are advantageous over other host cells, such as E. coli and yeasts, due to their high capacity for post-translational modifications, which are important for the proper folding and activity of antibodies. The cells are grown in large bioreactors and are fed with necessary nutrients and growth factors. Then, the antibodies are purified from culture media with various available purification techniques and are ready for use in in vitro, preclinical, and clinical studies.
Future perspectives
Antibodies have become one of the most successful classes of therapeutics used to treat various diseases, including cancer, autoimmune diseases, and infectious diseases. They are also essential tools for biomarker detection in the process of drug discovery.
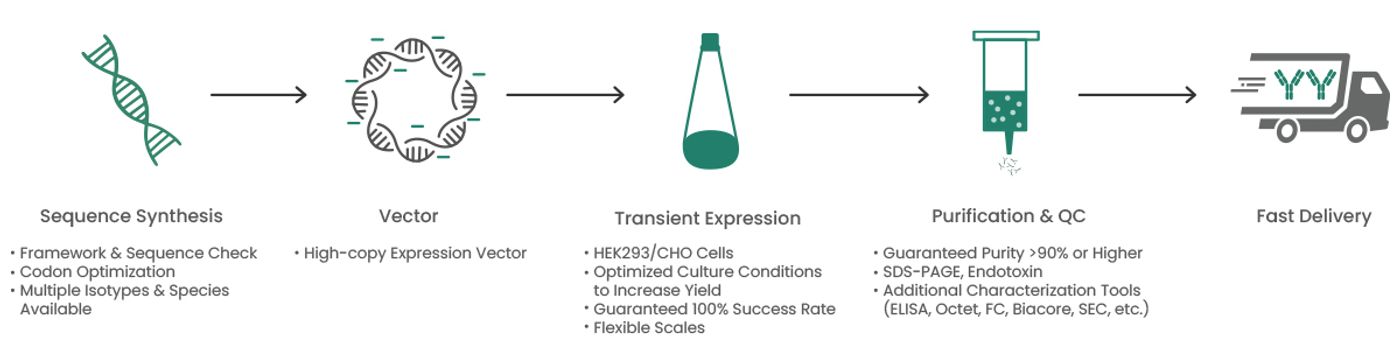
Researchers are still working on improving the manufacturing process and developing new ways of engineering and high-throughput (HTP) procedures to produce antibodies that can be used during drug discovery. Sino Biological is continually working on developing new workflows and methods to improve the efficiency and reduce the cost of antibody production. Sino Biological currently offers HTP recombinant antibody production service, providing the most cost-effective solution for the rapid production of a large number of antibodies. In response to the rapidly increasing therapeutic needs, Sino Biological combines its expertise in gene synthesis, vector design, and transient antibody expression technology to produce highly efficient antibodies in HEK293 and CHO cells in as fast as 2 weeks (Figure 6).
The future of recombinant antibody production is expected to be driven by several factors, including the development of new technologies, increased demand for therapeutics, and need for more affordable and efficient production methods. The field is likely to continue to evolve and expand, providing new opportunities for the development of more effective and accessible therapeutics.
References
1. Marks L. The birth pangs of monoclonal antibody therapeutics: the failure and legacy of Centoxin. MAbs. 2012;4(3):403-412. doi:10.4161/mabs.19909
2. Liu JK. The history of monoclonal antibody development - Progress, remaining challenges and future innovations. Ann Med Surg (Lond). 2014;3(4):113-116. Published 2014 Sep 11. doi:10.1016/j.amsu.2014.09.001
3. Geyer CR, McCafferty J, Dübel S, Bradbury AR, Sidhu SS. Recombinant antibodies and in vitro selection technologies. Methods Mol Biol. 2012;901:11-32. doi:10.1007/978-1-61779-931-0_2
4. Wang SS, Yan YS, Ho K. US FDA-approved therapeutic antibodies with high-concentration formulation: summaries and perspectives. Antib Ther. 2021;4(4):262-272. Published 2021 Nov 18. doi:10.1093/abt/tbab027
5. The Antibody Society. Therapeutic monoclonal antibodies approved or in regulatory review. (Accessed on January 10, 2023); www.antibodysociety.org/antibody-therapeutics-product-data