Natural Antibiotic Effective for Drug-Resistant Tuberculosis

A paper published in journal Molecular Cell, describes a naturally occurring antibiotic known as kanglemycin A which is related to the antibiotic rifampicin and is effective against Mycobacterium tuberculosis, the bacterium responsible for tuberculosis even in drug-resistant strains. "Rifampicin is already part of the cocktail of antibiotics used to treat tuberculosis, but many strains of the tuberculosis-causing bacteria have developed resistance to it,” says Katsuhiko Murakami, a professor of biochemistry and molecular biology at Penn State. "Tuberculosis is the leading cause of death by infectious disease worldwide. Development of rifampicin resistance in M. tuberculosis has made treatment of this disease very difficult since it extends the treatment time of tuberculosis from 6 months to 2 years. Identifying new compounds that are effective against the rifampicin-resistant bacteria is incredibly important for public health."
The researchers tested a library of naturally occurring compounds that have the ability to inhibit bacterial cell growth or prevent the production of RNA. They found that kanglemycin A was able to inhibit RNA production even in rifampicin-resistant bacteria. "Kanglemycin A is related to rifampicin, an antibiotic that functions by binding to bacterial RNA polymerase, the enzyme responsible for RNA production, and preventing it from making more RNA," said Murakami. "Understanding how kanglemycin A manages to maintain its affinity to rifampicin-resistant RNA polymerase and stay active against the drug-resistant bacteria will help to accelerate its approval for use in patients with tuberculosis."
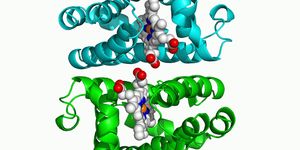
X-ray crystallography was then used by researchers to determine the pathway responsible for kanglemycin A and its actions against rifampicin-resistant RNA polymerase. "The X-ray structure actually revealed that kanglemycin A has two modifications that improve its function compared to rifampicin," said Murakami. "First, one of the modifications allows it to bind just outside of the rifampicin binding pocket increasing the strength of its affinity to the RNA polymerase in rifampicin-resistant bacteria. Second, another modification actually allows kanglemycin A to stop the synthesis of RNA even earlier than rifampicin."
"The previously unknown interactions of the unique chemical groups of kanglemycin A with RNA polymerase will direct the development of antibiotics against rifampicin-resistant M. tuberculosis. Approximately one third of the world's population is already infected with M. tuberculosis, and 600,000 people every year are diagnosed with rifampicin-resistant tuberculosis. Our work is the first step in developing a new drug for the treatment of these patients,” says Nikolay Zenkin, professor of molecular biology at Newcastle University in the U.K.
-
MAY 07, 2024Is It Anti-RNP or Anti-Sm/RNP?
- See More
-
APR 30, 2024Immuno-Oncology Virtual Event Series 2024
-
MAY 07, 20243rd International Biosecurity Virtual Symposium
-
MAY 23, 2024For the Love of Digital PCR 2024
- See More