Rapamycin was considered a wonder drug upon its discovery three decades ago when it was isolated from soil bacteria. It works as an immuno-suppressor in a variety of ways and has been to reduce immune responses in patients who received organ transplants. Now, rapamycin is garnering popularity again for its potential role in cancer therapy, neuroprotection, and most notably in anti-aging therapies.
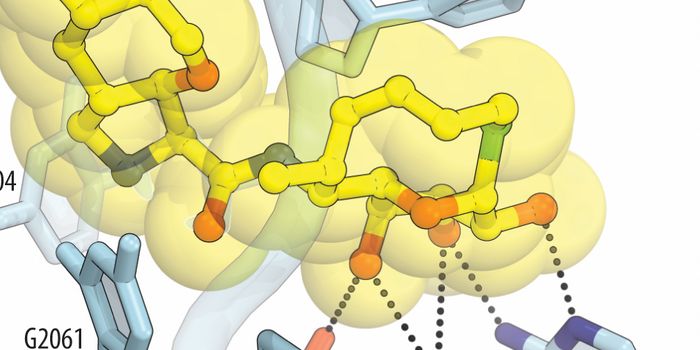
Rapamycin inhibits cell growth by targeting mTOR--the master regulator of cell growth in our cells. The targeting of mTOR leads to autophagy, a process by which cellular lysosomes eradicate misfolded proteins and damaged organelles.
"The main function of the lysosome is to maintain the healthy state of the cell because it degrades the harmful stuff within the cell," said Xiaoli Zhang, a postdoctoral researcher in the University of Michigan Department of Molecular, Cellular and Developmental Biology. "During stress conditions, autophagy can lead to the cell survival by degrading dysfunctional components and providing the building blocks of cells, such as amino acids and lipids."
Now, new research points to more than one cellular pathway being targeted by the rapamycin such pathway involves a calcium ion channel on the lysosomal membrane known as TRPML1 and is critical in regulating lysosomal function. "Without this channel, you get neurodegeneration," said Haoxing Xu, principal investigator of the study. "If you stimulate the channel, it's anti-neurodegeneration."
Findings were published in PLOS Biology.
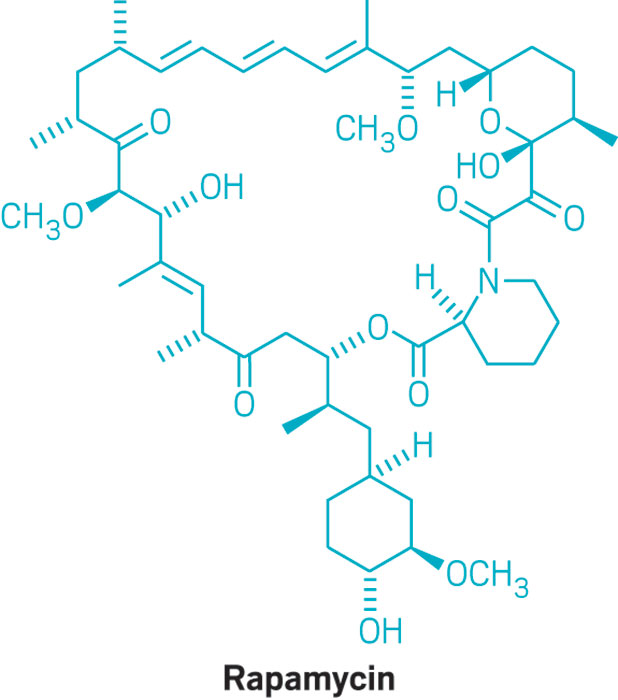
"We think lysosomal TRPML1 may contribute significantly to the neuroprotective and anti-aging effects of rapamycin," says lead author Wei Chen, who worked on investigating TRPML1 by using a unique technique called a lysosome patch clamp. "The identification of a new target of rapamycin offers an insight in developing the next generation of rapamycin, which will have a more specific effect on neurodegenerative disease."
Source: Medical Xpress

-
-
MAY 23, 2024For the Love of Digital PCR 2024
-
JUN 06, 2024The Future of Scientific Conferencing
- See More