Progress in the Development of Gene Therapy for Diseased Kidneys
Gene therapy has had an uneven trajectory after early missteps. But last year, the government began granting approval once again for using genetic manipulation to correct diseases. Scientists at the Washington University School of Medicine in St. Louis have now shown that genetic material can be effectively used to disrupted kidney cells in a mouse model. It may, therefore, be possible to treat chronic kidney disease. Roughly 30 million Americans are impacted by that often undiagnosed disorder, and there are few therapeutic options. This work has been reported in the Journal of the American Society of Nephrology.
"Chronic kidney disease is an enormous and growing problem," said senior author Benjamin D. Humphreys, MD, Ph.D., director of the Division of Nephrology at Washington University. "Unfortunately, over the years, we haven't developed more effective drugs for the condition, and this reality is leading us to explore gene therapy."
Chronic kidney disease sets in when the kidneys can no longer effectively remove waste and excess fluid from blood; it can cause hypertension, diabetes, and other illnesses as well. The symptoms - nausea, vomiting, swollen limbs, and sleep disturbances - are not specific enough for the disease to be easily diagnosed. Irreparable damage happens in the organ before it is identified, and it can also lead to heart disease.
"Part of the reason there have been so few advances in kidney disease treatment is because the kidney is complex, and we don't fully understand the disease process," explained Humphreys, the Joseph Friedman Professor of Renal Diseases in Medicine. "However, scientists are making progress, and I am optimistic."
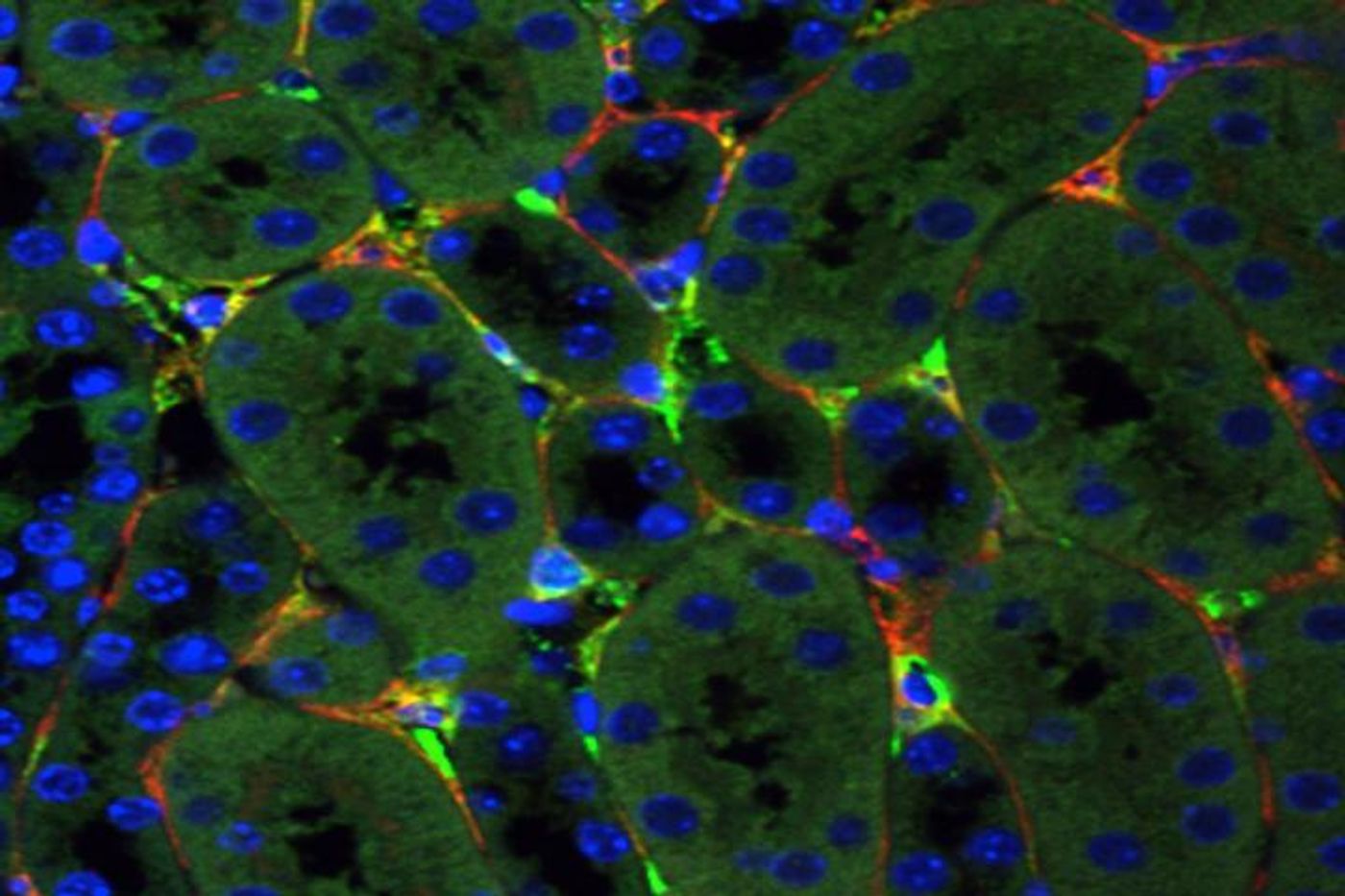
To try to develop a treatment, Humphreys and colleagues turned to adeno-associated virus (AAV), which is related to the common cold virus, as a delivery system for gene therapeutics. Before this work, no one had successfully targeted the organ and delivered genetic material there. This work shows it is possible.
Six AAV viruses were evaluated in both mice and kidney organoids - simplified models of the kidney that can be grown in the lab. Both synthetic and natural viruses were assessed, and in the end, Anc80, a synthetic virus engineered by a member of the research team successfully reached two different cell types in the kidney. Proteins released by those cells can harm the kidney. Anc80 was shown to be capable of transferring genetic material to them, and was tested as a treatment for kidney scarring in mice.
"This was a happy surprise," Humphreys said. "We were not expecting this."
This work is still in the early stages, cautioned Humphreys. The researchers still have to find the right genes to target so the disease can be corrected, a major hurdle. They also have to be sure that the virus will only act upon kidney cells.
"The interesting thing about the adeno-associated viruses is that they persist in the body for many months, potentially giving a therapeutic gene a chance to do its work," Humphreys noted. "Chronic kidney disease is a slowly progressive disease so that is an advantage. After many more years of research, we could envision that patients would need injections maybe twice a year as opposed to every week, like with chemo.
"There has been so little innovation in kidney treatment," he said. "We believe this is a positive step forward."
The video above illustrates how AAVs work.
Sources: AAAS/Eurekalert! Via Washington University in St. Louis, Journal of the American Society of Nephrology