CRISPR 2.0: Using Transposons to Improve and Expand CRISPR Application
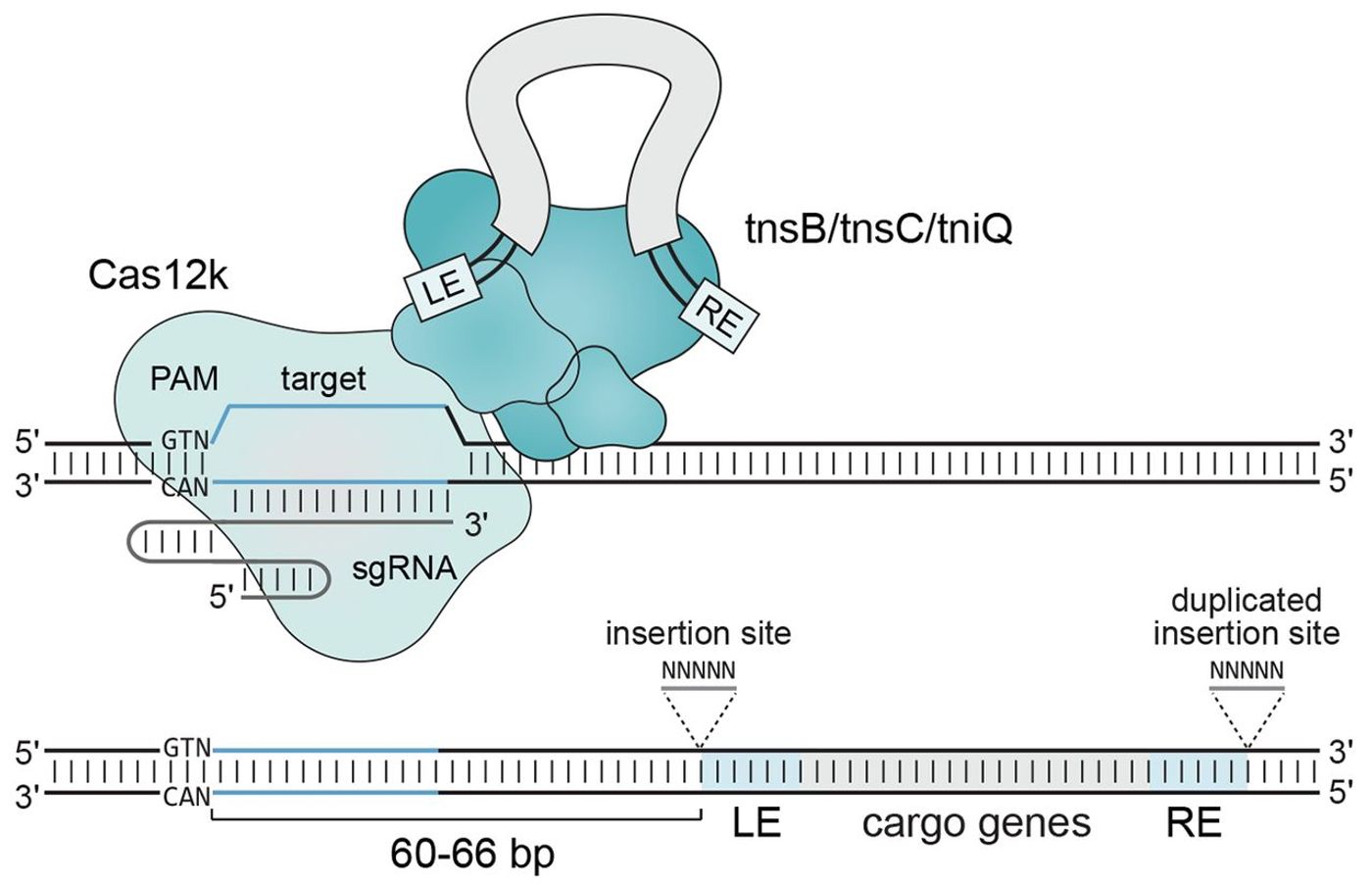
Source: Science showing cas12k binging and gene insertion.
CRISPR-cas9, commonly referred to simply as CRISPR, is a bacterial immune system that was first used as a gene-editing tool in 2012. Since the first application to gene-editing, CRISPR has made gene-editing faster, easy to design, and easy to execute. Resistance to CRISPR in human gene therapy is due to the risk of "off-target" effects. Though the risk is small, and debated, off-target effects can be devastating and irreversible. Additionally, CRISPR is very useful in deleting or inactivating genes, but scientists have found it very difficult to use it to insert or repair genes. While studies that demonstrate CRISPR's application continues, some scientists like Feng Zhang at the Broad Institute, have focused on making CRISPR more precise.
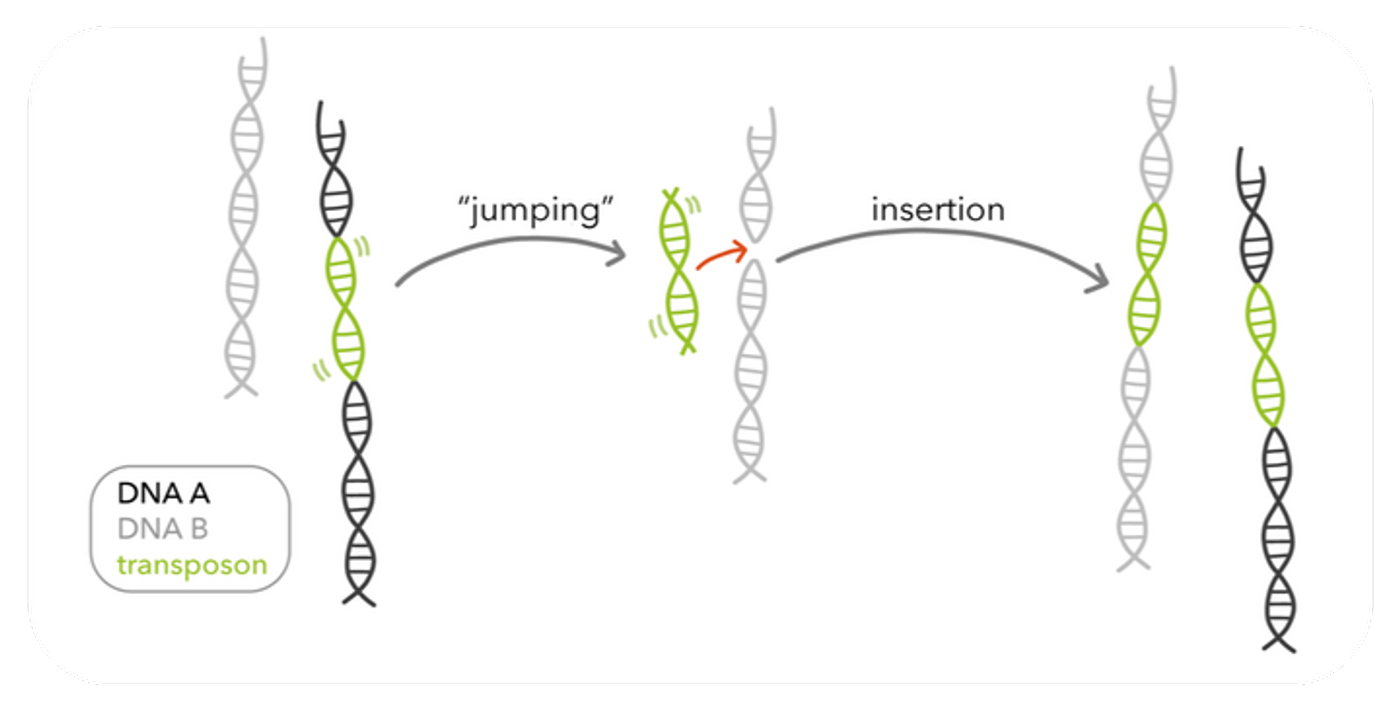
A team of scientists from the Broad Institute and the National Institutes of Health have engineered a CRISPR system which makes use of transposon activity to increase the specificity of gene-insertion and decrease the risk of off-target effects. The new mechanism is a sandwich with a shCAST loci in the middle, a transposase gene at one end, and a cas12k protein at the other. The shCAST locus is a type of CRISPR system which was isolated from a cyanobacterium. The job of shCAST is to integrate DNA at a specific site determined by the transposase. The transposase gene is called Tn-7, and like all transposons (or, "jumping genes") it can move around the genome. Tn-7 copies and pastes the gene it is attached to it in the engineered sandwich. Tn-7 precisely identifies the insertion site 60-66bp downstream of the protospacer sequence in shCAST. Lastly, cas12k is a protein sequence with the function of finding the protospacer sequence for Tn-7. All together, cas12k finds the protosequence, Tn-7 moves 60-66bp downstream of that sequence and then makes copies of the gene intended for insertion. Finally, shCAST inserts copies of the gene that Tn-7 synthesized.
Source: Harvard showing a basic schematic of transposons.
This new gene-editing technology has a successful insertion rate of 80% but has only been tested in bacterial organisms. Extensive experimentation in eukaryotic cells is needed before the technology can be considered for medical application. Still, finding possible solutions to the risks of CRISPR, only seven years after the groundbreaking technology was first used as a gene-editing tool, is exciting.
Sources: Discover Magazine, Science, GenomeWeb, NewScientist, Harvard