Optogenetic Techniques Provide Insight Into ALS
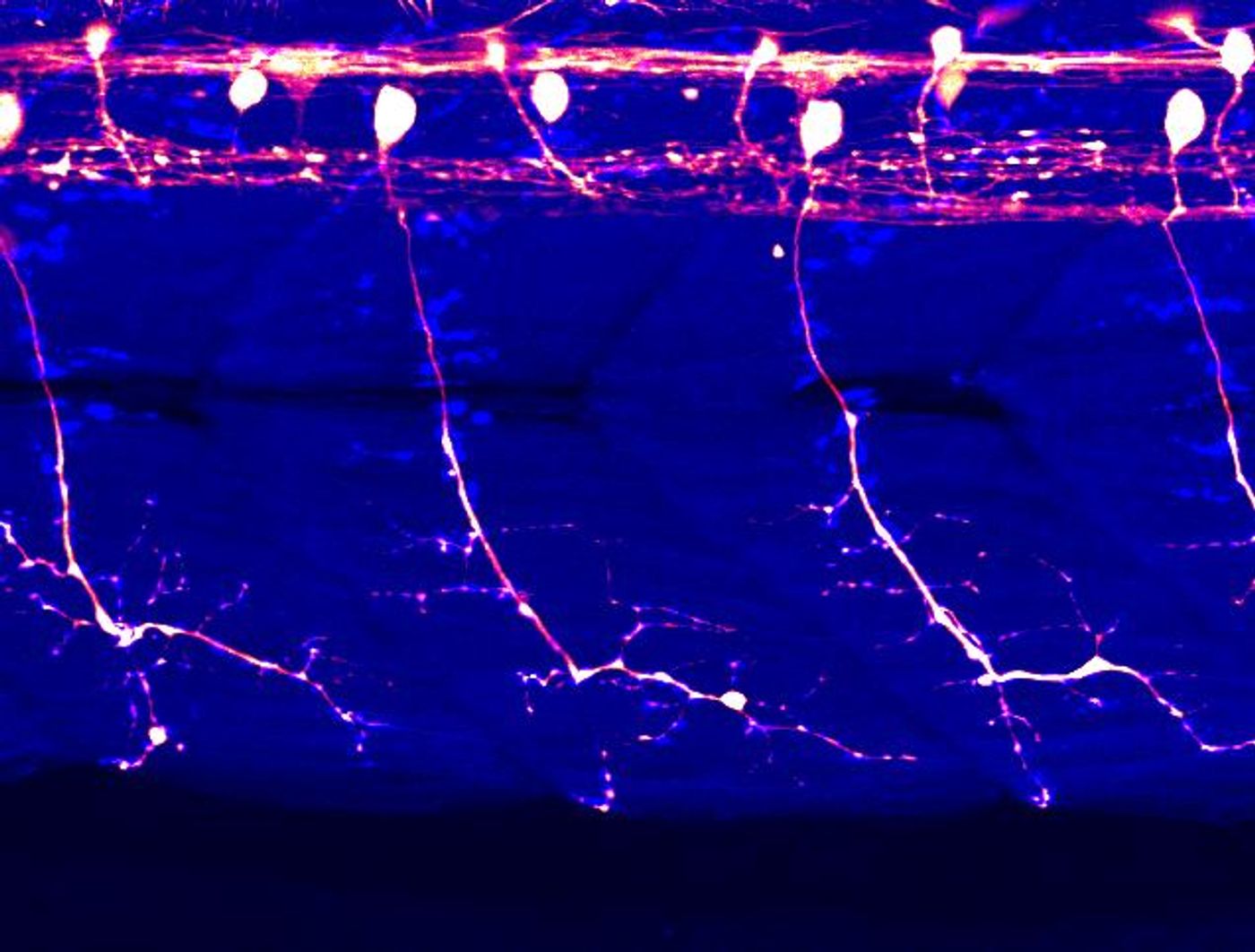
In humans, motor neurons link thoughts with the motion of the body. The bodies of these cells sit in the brain, brainstem, or spinal cord, and a projection from a motor neuron called the axon extends to muscles or organs. Signals are sent from the brain or spinal cord down to the muscles or organs that are innervated, causing a reaction or movement. This complex connectivity also also make motor neurons difficult to study.
In ALS (amyotrophic lateral sclerosis), the motor neurons begin to degenerate, and as they die, the brain has trouble triggering movement. People gradually lose physical abilities, and over time, cannot even speak, eat, or breathe. Since motor neurons are so challenging to observe, there is little we know about how neurons start to become dysfunctional in ALS.
Researchers have now been able to recapitulate the physical features of ALS in a common research model organism, the zebrafish. They used a technique that enabled them to manipulate the genetics of the zebrafish using light, called optogenetics.
Reporting in Nature Communications, the scientists engineered a variant of a protein called TDP-43, which is linked to ALS, that will function normally in the dark but will start to aggregate when exposed to blue light. In ALS, motor neurons build up inclusions that contain TDP-43 aggregates.
In the video below, a zebrafish carrying the optogenetic TDP-43 was exposed to blue light, and the fish began to show ALS-like features. The links between motor neurons and muscles began to get weaker even after the fish were not being exposed to light, and prior to the aggregation of TDP-43. These findings suggest that motor neurons are degenerating before the large buildup of TDP-43 can be observed; they are often seen in the terminal stage of the disease.
"This research, for the first time, shows that TDP-43 aggregation is a cause of motor neuron dysfunction in animals. We think that the small TDP-43 assemblies, which are called TDP-43 oligomers, might be more toxic to motor neurons than the larger aggregates," said Dr. Kazuhide Asakawa, who spearheaded this research.
"We can now produce an ALS-like state in a temporary and spatially tuned manner by controlling light intensity and the position of illumination. Our ultimate goal over the next few years is to identify chemicals that prevent optogenetic TDP-43 from forming oligomers and aggregates, and we hope such chemicals will be used for ALS treatment," he added.
Sources: AAAS/Eurekalert! via Research Organization of Information and Systems, Nature Communications