How Autism-Associated Mutations in One Gene Impact the Brain
Autism spectrum disorder is complex; it presents differently in different patients and may be influenced by many factors, some of which are genetic and others that may be environmental. But there are some cases in which a gene has been definitively connected to ASD. The Cullin3 gene is one example; mutations in this gene cause ASD in carriers, and now scientists have learned more about why. Though mutations in Cullin3 are thought to be relatively rare, this study may tell us more about the biological basis of ASD. The findings, which have been reported in Nature Communications, relied on a mouse model that carried Cullin3 mutations.
Several types of behavioral and movement tests with the mouse model suggested that they mimicked some features of autism, such as reduced sociability and motor coordination defects.
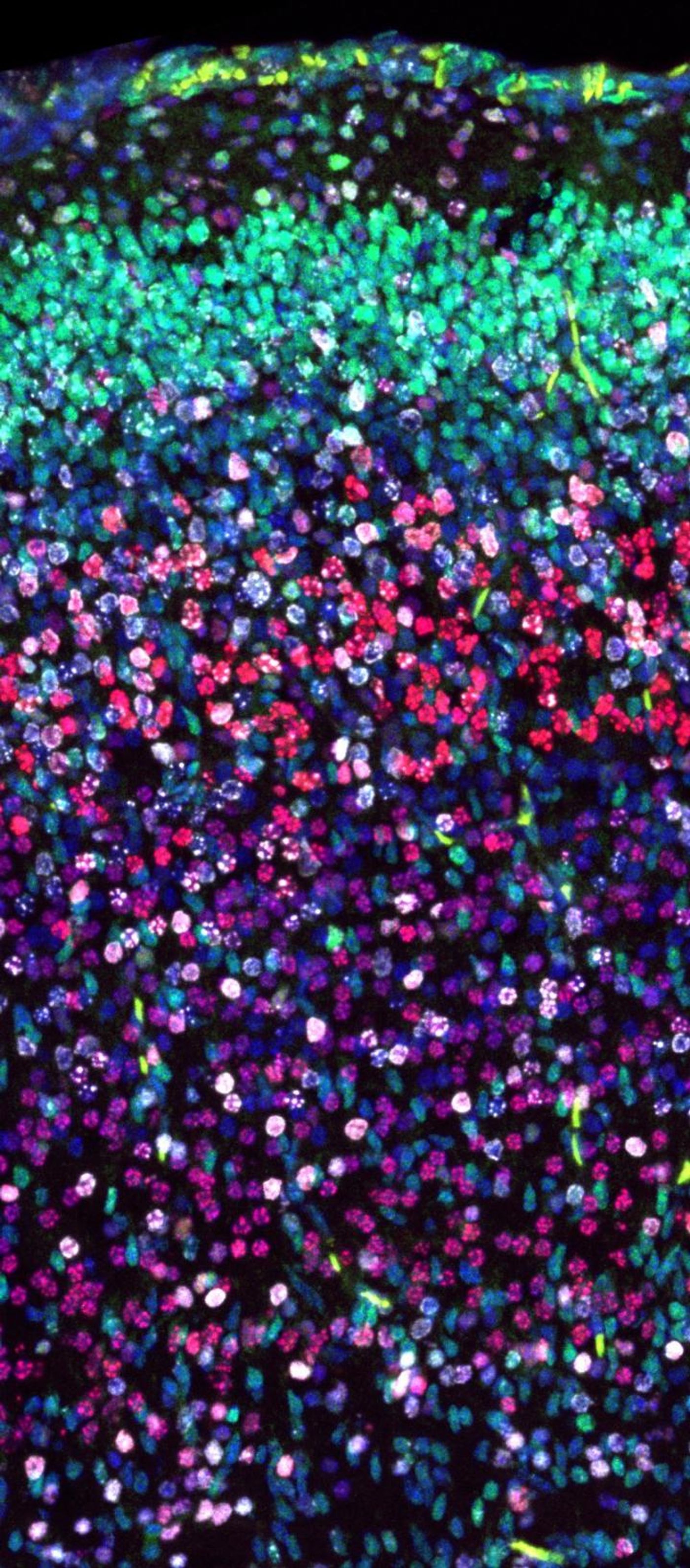
The researchers then began to investigate the biology of the mouse model, and found that in a very small subset of neurons, the migration and growth was altered. Neurons normally originate from a specific place and then move up through various layers of the brain until they reach their correct final location in the cortex. This is a fine-tuned process that's extremely vulnerable to even small changes.
The scientists tagged the neurons in the mouse model so they could be visualized as they migrated, and found that the process was disrupted. "We could observe migration deficits - the neurons are stranded in the lower cortex layers," said co-first study author and graduate student Lena Schwarz.
The Cullin3 gene encodes for a protein that tags other proteins when they are degraded and ready for disposal. Dysfunctional proteins can't be allowed to accumulate in cells or they'll cause problems. The researchers began a systematic study to reveal which proteins were dysregulated when Cullin3 was mutated.
"We were looking at proteins that accumulate in the mutant brain and found a protein called Plastin3. Then [Professor Gaia Novarino] came across a poster describing the work of IST Austria's Schur group in the hallway, and we got very excited," said co-first study author Jasmin Morandell, Ph.D.
"They independently had been working on Plastin3 as a regulator of cell motility and had complementary results to ours. That's when we started working together," added study supervisor Professor Gaia Novarino.
Plastin3 was not known to be involved with the migration of neurons, but this work has changed that. "If the Cullin3 gene is deactivated, the Plastin3 protein accumulates, causing cells to migrate slower and over shorter distances. This is exactly what we saw happening in the cortex of the Cullin3 mutant mice," Schwarz explained.
This neural migration occurs very early on in development, and would not be detectable in a fetus with current technologies.
"Determining these critical windows during brain development could be extremely important to fine-tune the treatment of patients with specific forms of ASD," Novarino added. "Following up with the research on Plastin3 could pave the way for some therapeutics. Inhibiting the accumulation of this protein could eventually alleviate some of the symptoms patients have," Schwarz said.
"We now know that defective Cullin3 leads to increased levels of Plastin3. This tight correlation shows that Plastin3 protein levels may be an important factor for the control of cell-intrinsic movements," said Morandell.
Sources: AAAS/Eurekalert! via Institute of Science and Technology Austria, Nature Communications