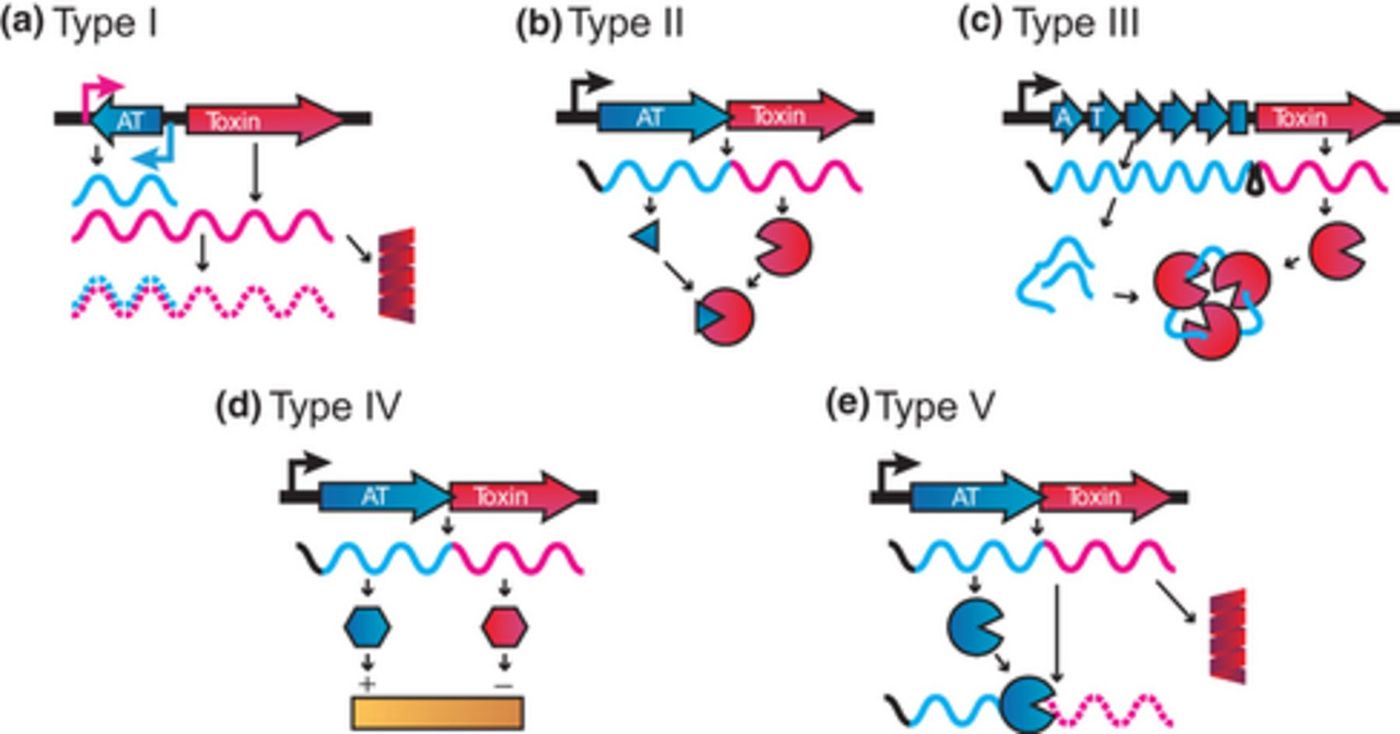
Bacteria are not without protection from antibiotics; they actually incorporate a system of closely-linked genes from the same chromosome coding for proteins acting as “poisons” and “antidotes.” New findings from the University of Geneva describe a way to use bacteria’s security system against them, potentially boosting the treatments of various bacterial infections and reducing dependency on antibiotics.
Under normal conditions where bacteria do not observe any threats to their vitality, the antidote protein inhibits the toxin from killing the cell. However, during environmentally stressful situations like exposure to antibiotics, a third party breaks down the antidote proteins so nothing stops the toxin from causing cell death.
Researchers from the University of Geneva looked specifically at the toxin-antitoxin system in
Caulobacter crescentus, called HigBA. Although HigBA is found in many pathogenic and non-pathogenic bacterial species, it is unlike some bacterial toxin-antitoxin systems in a few ways that make it easier to manipulate in the lab.
"Normally, toxin activation puts the cells into hibernation by shutting down their basic functions, allowing them to reactivate later on,” said Clare Kirkpatrick, first author of the study. "HigBA, on the contrary, is highly specific both in its activation conditions and its response. It is dedicated exclusively to the DNA damage response in these bacteria and attacks a small set of essential targets in the cell, leading to inescapable cell death."
Manipulating some toxin-antitoxin system are dangerous for this reason. For those bacteria that are not killed by their internal toxin and are instead sent into a dormant state to avoid destruction by antibiotics, one they return from their dormant state, they could be more resistant and more powerful than ever.
Artificially inactivating the antitoxin gene in
C. crescentus HigBA to induce cell death was especially easy because of a unique system of regulation surrounding HigBA, although HigBA is not the only toxin-antitoxin system that incorporates a similar regulatory pathway.
In addition to the HigBA antitoxin acting as a repressive transcription factor that prevents expression of both the toxin and antitoxin genes, the team from the University of Geneva found a second repressor. Even more, the second repressor was stricter in its prevention of gene copy production for the toxin and antitoxin genes.
The second repressor only responds to stress from damage to the bacterial DNA, like when antibiotics are administered. Additionally, the second repressor “permits the mutation of the antitoxin gene.”
"Our discovery can change the way we fight bacterial infections. Instead of using chemical warfare, i.e. antibiotic unspecifically, we could force bacteria to turn their weapons on themselves by treating bacteria with selected combinations of antibiotics," said team leader Patrick Viollier.
As scientists cautiously continue to study manipulation of bacterial defense systems, a new age of biocontrol could ensue. However, bacteria manage to fight back against antibiotics. What will be their response to scientists’ genetic meddling?
Source:
University of Geneva