Obesity, diabetes, and other metabolic conditions are a huge problem for which there is not yet an effective solution. Annual healthcare costs related to obesity are about 200 billion dollars (~20% of all healthcare costs). Most people know that muscle uses more energy at rest than adipose tissue (fat) does, but a lot of people don’t know about brown adipose tissue. Similar to muscle, brown adipose tissue (BAT) is a thermogenic organ, meaning it burns more energy at rest. Being able to pharmacologically manipulate BAT to burn more calories would be an effective way to combat obesity, but this has not yet been a viable strategy. BAT can be activated by stimulating β adrenergic receptors (βARs), but those same receptors are also heavily involved in cardiac function. In order to pharmacologically stimulate BAT to burn more energy without causing dangerous cardiac symptoms, a new, safer target is needed.
A recent paper published in
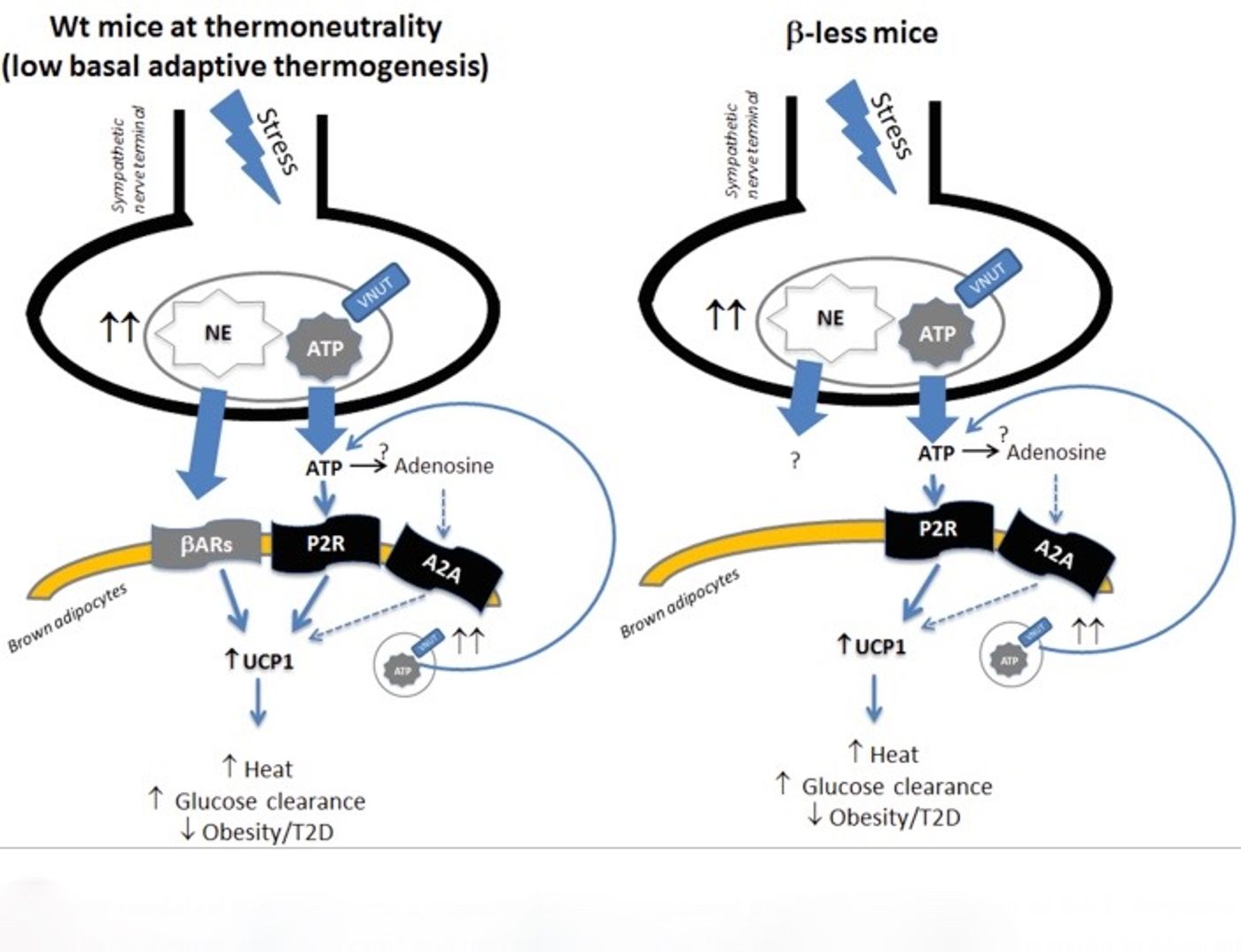
Molecular Metabolism investigated the effect that baseline BAT activity has on resilience to weight gain during chronic stress. Briefly, the results suggest that resilience to weight gain under conditions of chronic stress is determined by the state of BAT activity before the stress occurred. In mice, low adaptive BAT activity before stress provided resistance to weight gain, and higher baseline BAT activity pre-stress led to weight gain. Interestingly, all the mice tested exhibited similar levels of overeating, meaning the greater BAT activity seen during stress was able to overcome the excess energy intake due to overeating in the low adaptive BAT activity mice. This particular finding is not super translatable to humans because, to keep the mice in a state of low BAT activity, they were either βAR-knockout mice or were kept in a thermoneutral environment. Here, thermoneutral refers to the temperature at which an animal does not have to use energy to regulate its body temperature. Humans obviously have βARs and we generally live in thermoneutral comfort zones, but many of us still gain weight in response to chronic stress.
The most interesting, and most relevant, part of this paper is the pathway that the researchers described that controls this response in mice. The βAR-KO mice exhibited more BAT activity and a resistance to obesity under conditions of chronic stress, but the BAT could not have been stimulated through βARs. From that observation and previous work, they propose a model where BAT activation is at least partially controlled through a purinergic signaling pathway. Importantly, this pathway is conserved in humans so it could provide a new pharmacological target to activate BAT without causing serious side effects. Exposure to cold can also activate BAT, but the reality is that more people would probably choose a pill over hours in a cold room. Dealing with obesity and its related concerns is imperative, so hopefully this purinergic pathway will be targeted for drug development.