Eating Bacteria in Self-Defense: A strategy against tuberculosis
Cells throughout our body can defend themselves from bacteria using various mechanisms. By harnessing a cellular process called autophagy, researchers at The Crick hope to boost our body’s ability to protect against infections such as Mycobacterium Tuberculosis (TB).
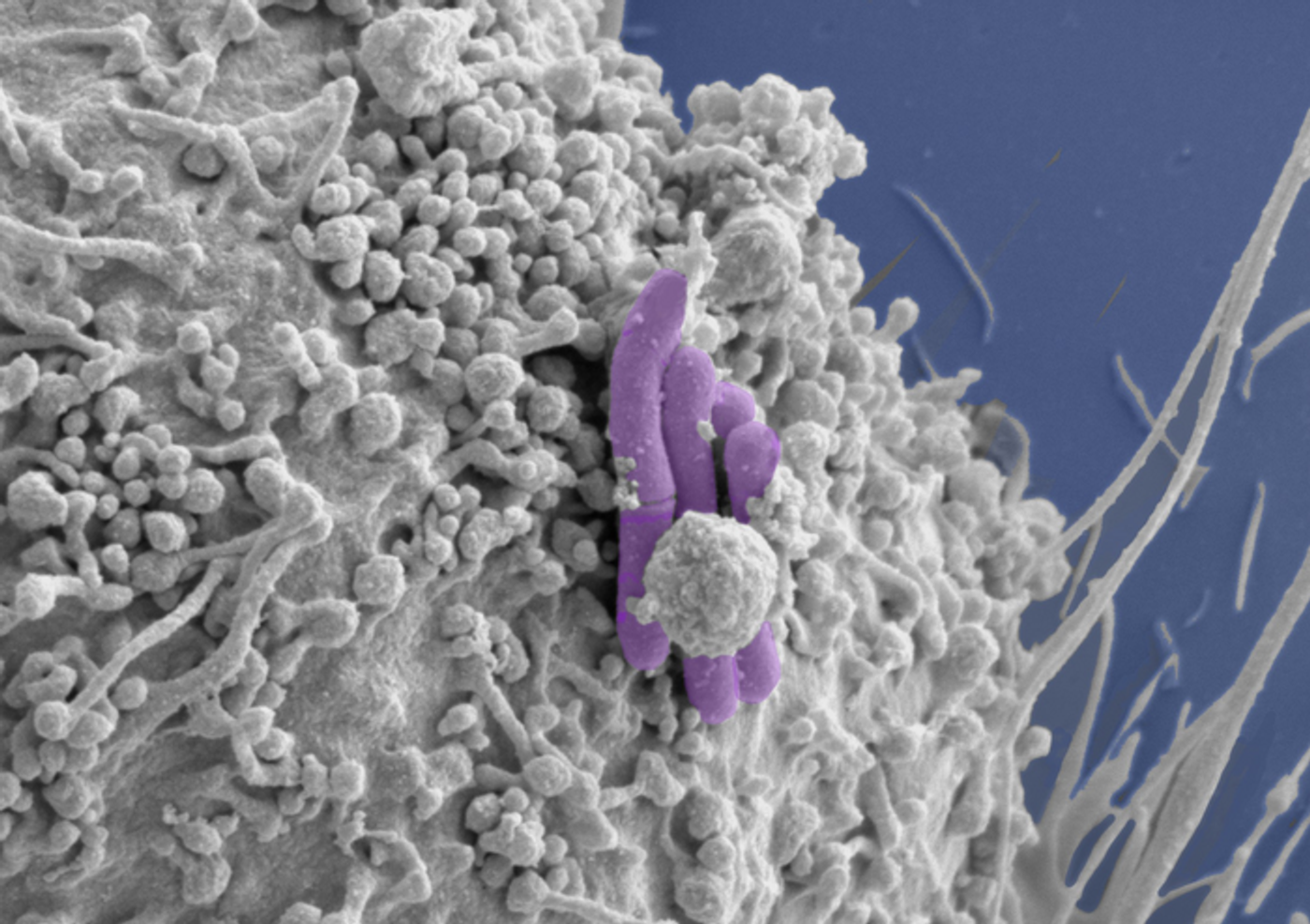
Image of a macrophage (grey) infected with M. tuberculosis (pseudocolored purple), the bacilli bacteria that cause TB. Image Credit Tony Fearns (Host Pathogen Interactions in Tuberculosis Lab)
For 150 million years, animals on earth have been literally plagued with tuberculosis. Today, around 13 million people in the United States are living with a TB infection. TB is a vital infection to study, according to the co-first author of a recent Nature Microbiology article, Beren Aylan. “It takes a very long course of different antibiotic treatments to effectively remove the infection,” explains Aylan. “Anything that can be done to more effectively remove bacteria, could also make a huge difference to the cost and accessibility of treatments.”
One of the many ways TB evades and invades is by dodging a cell’s autophagy process. Autophagy is how a cell cleans up its cluttered cellular compartment or produces quick energy by “eating” its less essential components. Cells under extreme stress or infection can enact self-destruction using autophagy. Previous studies have revealed this mechanism functions as a form of self-defense against bacterial infection.
In their triple first-authored paper, Aylan, Bernard, Pellegrino et al. conducted experiments on human macrophages they derived from induced pluripotent stem cells (iPSDM).
The researchers turned various autophagy-related genes off in these iPSDMs and recorded how well the cells resisted TB infection. In tandem, various mutated versions of TB were tested on their ability to trigger autophagy.
Max Gutierrez PhD. and senior author from the Host-Pathogen Interactions in Tuberculosis Laboratory at the Crick reports by “using the latest technologies, we’ve been able to show a key role for this pathway in controlling infection.”
The researchers identified genes associated with autophagy that were critical for TB defense. Knocking out these genes led to TB infection, replication, and TB-related cell death.
By artificially regulating autophagy, these researchers suggest current antibiotics could be improved. Alternatively, further research could develop new treatments for antibiotic-resistant bacterial strains. With the threat of antibiotic resistance looming, co-first author, Beren Aylan believes “it’s incredibly important to understand how our bodies fight infection and where there might be room for improvement.”
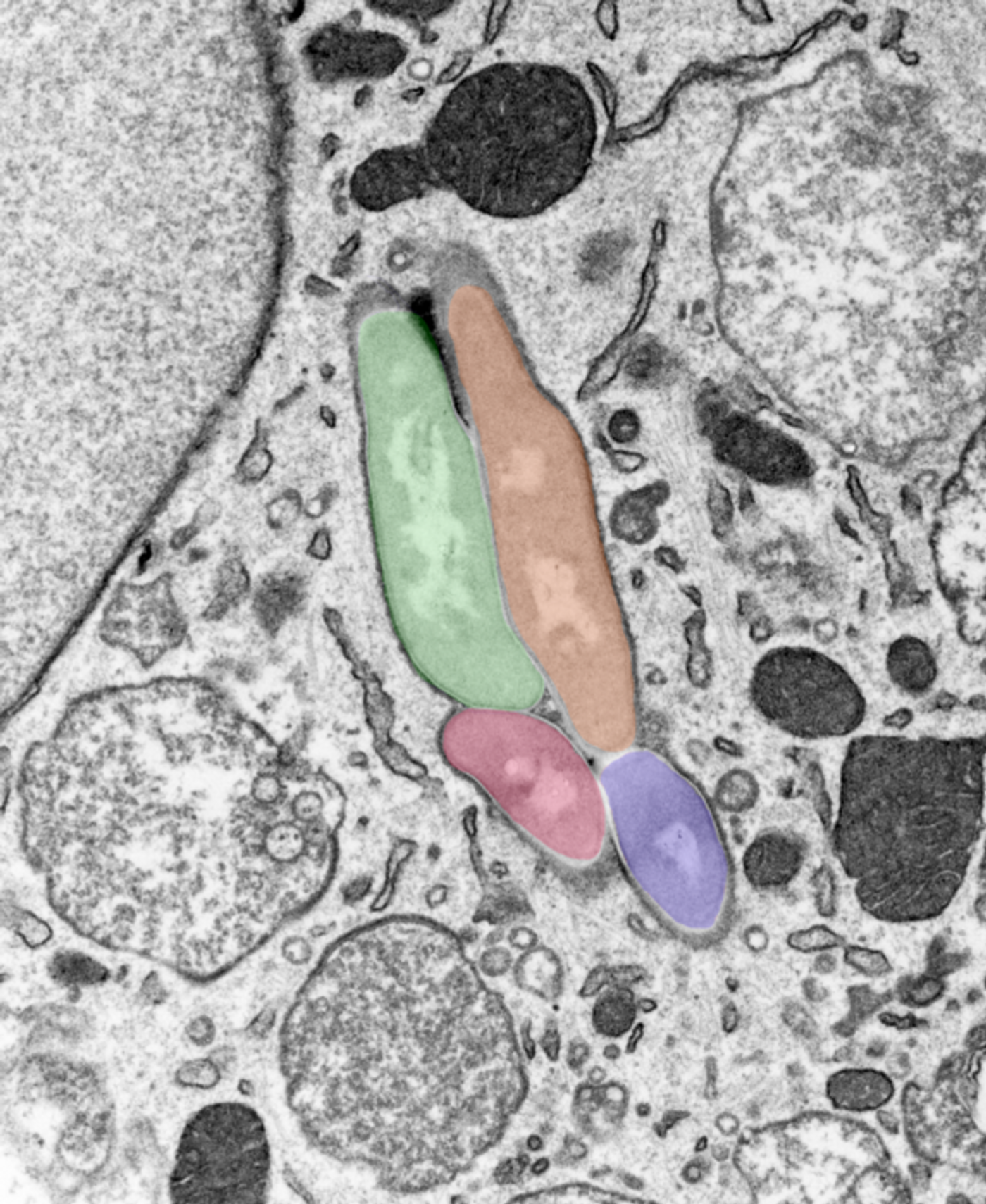
Image showing a macrophage infected with several M. tuberculosis bacilli (pseudocolored in different colours). Tony Fearns (Host Pathogen Interactions in Tuberculosis Lab)
Much like immunotherapies for cancer, the hope is that scientists could design treatments to support a person’s immune response against infection by boosting the autophagy pathway.
Previous research suggests bacteria can interfere with a host cell’s autophagy. However, this research stops short of indicating that boosting autophagy is possible or could decrease the success of bacterial infection.
Strengthening the cellular structures and mechanisms surrounding autophagy is the next challenge for these researchers. The hope is new drug compounds might aid our cell’s internal defense system by targeting crucial components of the autophagy pathway.
Sources: CDC, Nature Microbiology, Experimental & Molecular Medicine