A clinical trial at the Duke Translational Research Institute was immediately shut down after 10 participants had an anaphylactic immune response to a polymer used in the trial to extend the effectiveness of an anticoagulant drug. Scientists involved in the trial teamed up with other Duke researchers, and the product of their collaboration is a redesigned drug-delivery technology to prevent future immune responses.
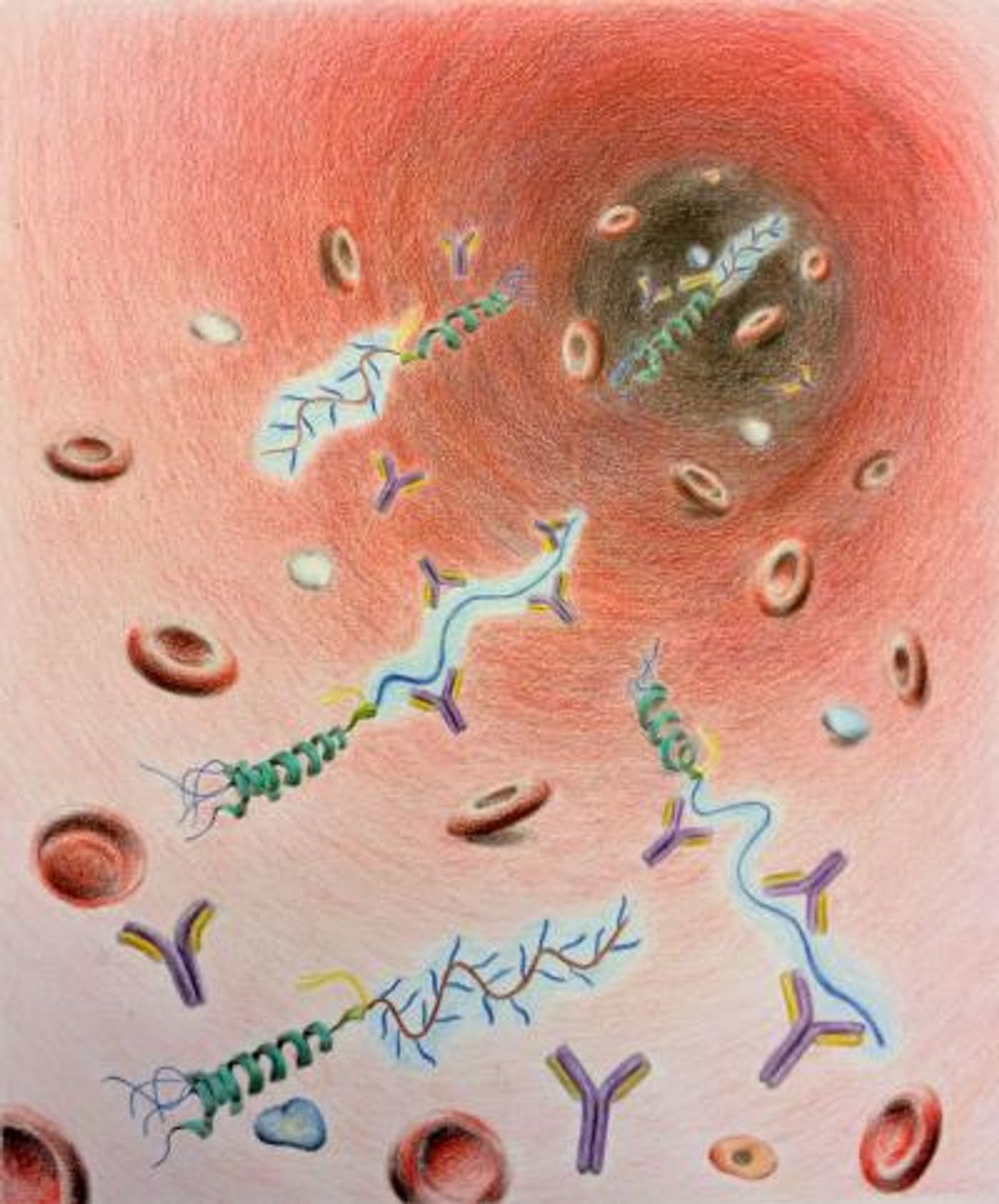
Polyethylene glycol (PEG) is the polymer at stake. It’s found in pharmaceuticals and in commercial products like toothpaste and cosmetics for the purpose of thickening, softening, and moisture-carrying. But lately PEG has been commonly used alongside active drugs administered to the bloodstream to prevent the body from immediately flushing out the drugs, boosting the amount of time the drugs can have an effect.
"Because people with pre-existing PEG antibodies are now present in the population and such antibodies can elicit life-threatening allergic reactions, it has become critical for drug development to create alternative formulations of PEG,” said Bruce Sullenger, director of the DTRI.
Because PEG is so often used, people have developed antibodies to PEG, from so-called “PEGylated” drugs, similarly to the way bacteria become resistant to commonly used antibiotics. In this situation, drugs lose their efficacy time and the PEG-attached drugs have the potential to trigger possibly dangerous allergic reactions.
The new research study prompted by the unexpected allergic reactions in the DTRI study led to an altered version of PEGylated drugs that prevents the recognition of antibodies already in existence in people’s bodies, although the group initially started their project for the purpose of boosting production efficiency of the new PEG delivery system.
"We started down a path to make PEG-like conjugates of protein drugs more efficiently and stumbled into the PEG antigenicity problem," said Ashutosh Chilkoti.
During mice experiments, the researchers successfully used the new drug delivery system to control glucose levels in mice with type 2 diabetes for up to four days, when the drug alone without PEG is only capable of controlling diabetes for six hours. The secret to the success might be that the new polymer technology involves “growing” PEG directly from a specific site on a drug molecule. According to researchers, this method prevents a labor-intensive process.
"Growing the polymer directly on the drug is simpler and more efficient in terms of yield than the conventional process," explained Yizhi Qi, a graduate student involved in the project. "The boost in efficiency varies from protein to protein, but our yield is significantly higher and produces more uniform results."
In future studies, Duke scientists plan on exploring the possibility of antibodies being creating against the new polymer technology. The present study was published in the journal
Nature Biomedical Engineering.
Source:
Duke University