Gut Bacteria Influence the Efficacy of a Parkinson's Drug
We all host a vast community of microbes in our gut, a part of the body that’s been called a second brain because of its close connections with our central nervous system. The bacteria in our digestive tract helps us metabolize food, and it can also impact oral therapeutics. New research by scientists at the University of Groningen has shown that gut microbes are interfering with levodopa, a treatment for Parkinson’s disease. Levodopa is an oral therapy that is meant to be changed into dopamine inside the brain; when the conversion happens before it gets there, the dopamine is not able to cross the blood-brain barrier. These findings have been reported in Nature Communications.
“It is well established that gut bacteria can affect the brain. There is a continuous chemical dialogue between gut bacteria and the brain, the so-called gut-brain axis,” explained the lead investigator of this study Sahar El Aidy, Assistant Professor in Microbiology.
Levodopa is a pill that is taken up by the small intestine and moved to the brain through the bloodstream. Enzymes called decarboxylases change the drug into dopamine before it reaches the brain. To prevent that from happening, patients take decarboxylase inhibitors.
“But the levels of levodopa that will reach the brain vary strongly among Parkinson's disease patients, and we questioned whether gut microbiota were playing a role in this difference,” noted El Aidy.
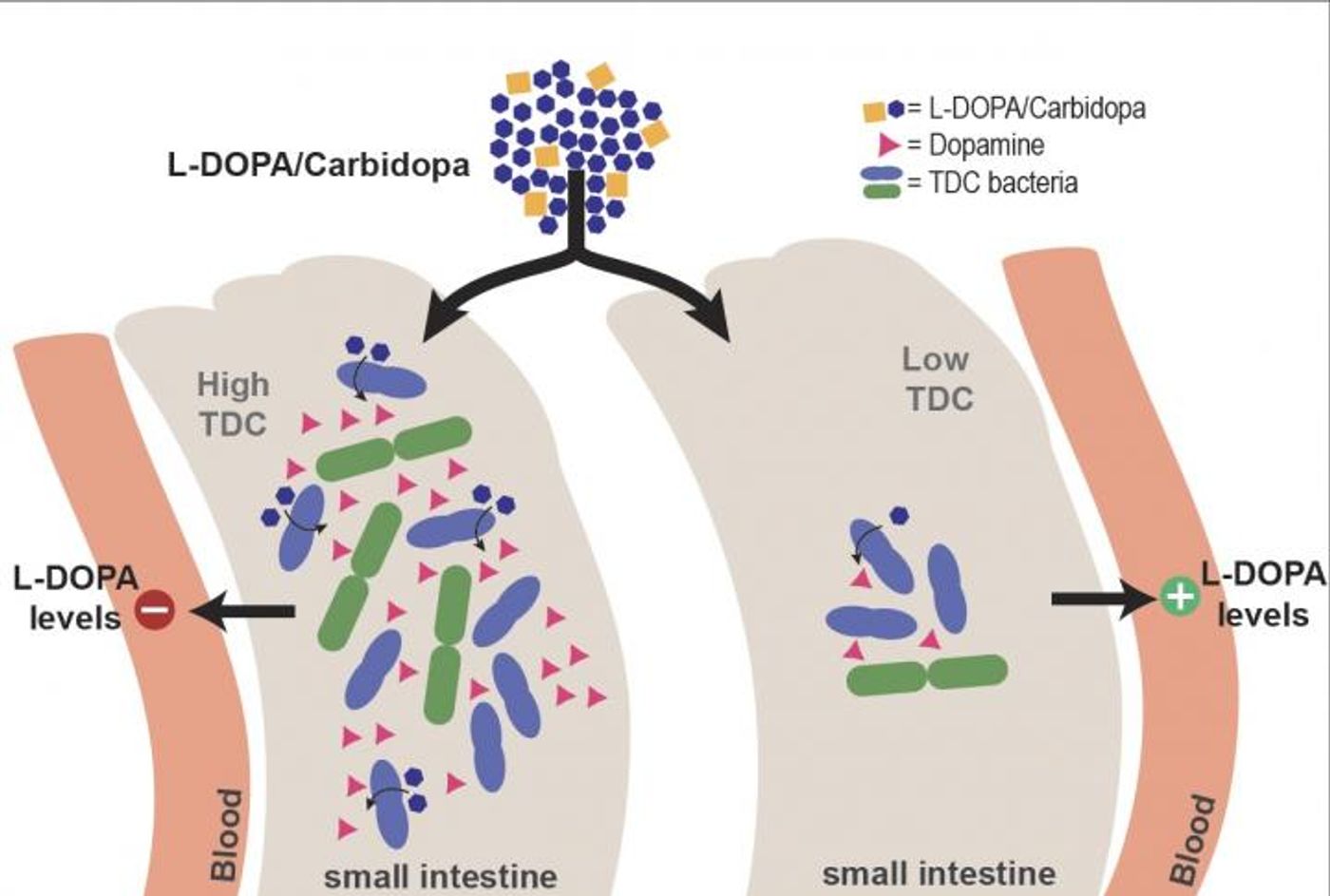
Graduate candidate Sebastiaan van Kessel used a rat model to assess the activity of a bacterial enzyme called tyrosine decarboxylase. The enzyme can convert levodopa into dopamine. The researchers showed that Enterococcus bacteria were producing that enzyme, and that even when it was required for its normal job, it continued to modify the levodopa.
Since patients that take this drug are also given decarboxylase inhibitors, the team assessed the effect of inhibitors on the bacterial enzyme. “It turned out that, for example, the inhibitor Carbidopa is over 10,000 times more potent in inhibiting the human decarboxylase,” revealed El Aidy.
The investigators wanted to see how all of this might be playing out in Parkinson’s patients and were able to obtain stool samples. That allowed them to assess how active the bacterial tyrosine decarboxylase enzyme was in these patients by measuring the activity of the gene encoding for the enzyme. Patients with higher gene activity levels were found to need higher drug doses.
“As these were stool samples, and the levodopa is absorbed in the small intestine, this was not yet solid proof. However, we confirmed our observation by showing that the higher abundance of bacterial enzyme in the small intestines of rats reduced levels of levodopa in the bloodstream,” noted El Aidy.
The team also learned that higher levels of the bacterial enzyme correlate with disease duration. Because of another drug used to treat gastrointestinal distress taken by Parkinson’s patients, an overgrowth of bacteria, including Enterococci, can develop in the small intestine. This can lead to a vicious cycle in which the symptoms of the disease only get worse.
El Aidy suggested that the bacterial decarboxylase levels can help explain why some Parkinson’s patients need more levodopa than others. “This is considered to be a problem for Parkinson's disease patients, because a higher dose will result in dyskinesia, one of the major side effects of levodopa treatment.”
Sources: AAAS/Eurekalert! via University of Groningen, Nature Communications