Alzheimer's Linked to a Gum Disease-associated Bacterium
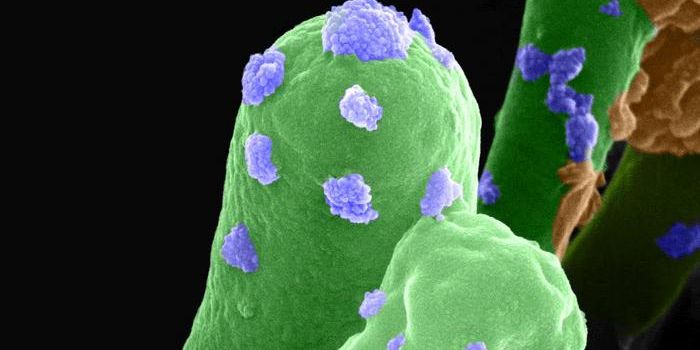
We know that as people age, Alzheimer’s disease becomes a more likely occurrence. While researchers have found some hallmark characteristics of the disorder, the exact causes are still unclear. Other than age, genes probably also play a role in the development of the illness, as do lifestyle choices and environmental influences. It’s even been suggested that microbial infections in the brain may be to blame, which seemed an unlikely hypothesis to many researchers. New work has indicated, however, that a bacterium associated with chronic gum disease, Porphyromonas gingivalis (Pg), appears to drive Alzheimer’s disease (AD).
New work reported in Science Advances by University of Louisville researcher Jan Potempa, Ph.D., of the Department of Oral Immunology and Infectious Diseases in the School of Dentistry, and others led by Cortexyme Inc., a private pharmaceutical company, has identified the Pg bacterium in patients with Alzheimer’s. Potempa believes their study is more compelling than previous research.
He commented that “we now have strong evidence connecting P. gingivalis and Alzheimer's pathogenesis, but more research needs to be done. An even more notable aspect of this study is demonstration of the potential for a class of molecule therapies targeting major virulence factors to change the trajectory of AD, which seems to be epidemiologically and clinically associated with periodontitis.”
In an animal model, Pg could live in the brain and increase the production of a molecule called amyloid beta. That molecule is a known component of an Alzheimer’s hallmark, amyloid plaques.
Related: Might Alzheimers Disease Stem From a Brain Infection?
The researchers also found toxic enzymes made by the microbe in Alzheimer’s patient neurons. These enzymes, gingipains, move to the outer surface of bacterial membranes and have been shown to regulate Pg toxicity in various cell types. Gingipain levels were correlated in this work to tau, which is necessary for the normal function of neurons, and ubiquitin, which the cell uses to tag damaged proteins.
Cortexyme sought a drug that could reduce the neurotoxicity caused by Pg, and targeted gingipains with small molecules. They found a compound that could reduce neuroinflammation and increase neuronal protection, lowering the damage caused by Pg infection in the brain. Last October, the company revealed that positive trends were seen in a clinical trial of this compound in Alzheimer’s patients. Phase 2 and 3 trials are now planned for 2019.
Sources: AAAS/Eurkealert! Via University of Louisville, Science Advances