Bacteria Exposed to Antibiotics are 'Primed' to Gain More Resistance
Antibiotics are critical drugs that have saved millions of lives, but bacteria can also gain resistance to them, rendering them less effective. The rise of antibiotic-resistant bacteria is recognized as a threat to public health, and while researchers know some of the reasons why it can be so easy for bacteria to become drug-resistant, it's something we're always learning more about. Scientists have now learned that when an infectious microbe is already resistant to a drug and is then exposed to that drug for a prolonged period of time, it holds onto the gene promoting resistance more tightly. Antibiotic-exposed resistant microbes are also more likely to acquire and maintain resistance to another antibiotic. The findings, which show how easy it is for a multi-drug resistant bacteria to evolve, have been reported in Nature Ecology & Evolution.
"Exposure to antibiotics appears to select indirectly for more stable antibiotic resistance systems," said co-senior study author Benjamin Kerr, a University of Washington (UW) professor of biology. "A more stable system in a strain will increase the chances that it will acquire resistance to multiple antibiotics."
When bacteria were exposed to an antibiotic for awhile, the bacteria were 'primed' to gain more antibiotic resistance, even after the antibiotic exposure stopped. This effect enabled the microbes to retain their resistance for generations.
"This could help explain not only the rise of multidrug resistance in bacteria, but also how antibiotic resistance persists and spreads in the environment -- in health care settings, in soil from agricultural runoff -- even long after the antibiotic exposure has ended," said the co-senior study author Eva Top, a professor of biology at the University of Idaho.
Resistance genes are often found on small, circular pieces of DNA called plasmids that can be easily passed from one microbe to another. They can also have deleterious effects on a microbe, and may be shed quickly.
"Even though they can carry beneficial genes, plasmids can also interfere with many types of processes inside a bacterial cell, such as metabolism or DNA replication," explained lead study author Hannah Jordt, a UW research scientist in biology. "So, scientists have generally thought of plasmids as costly and burdensome to the host cell."
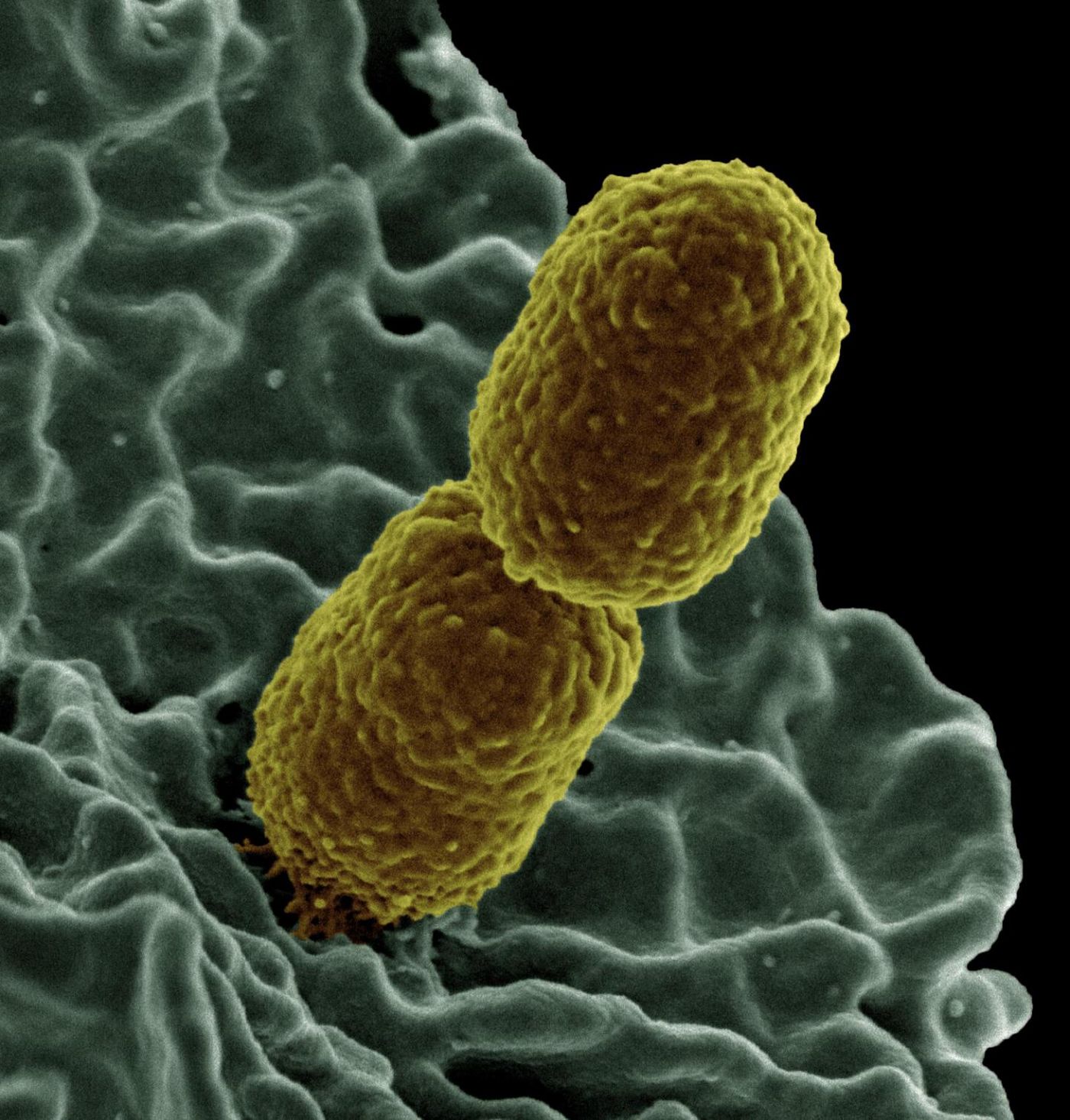
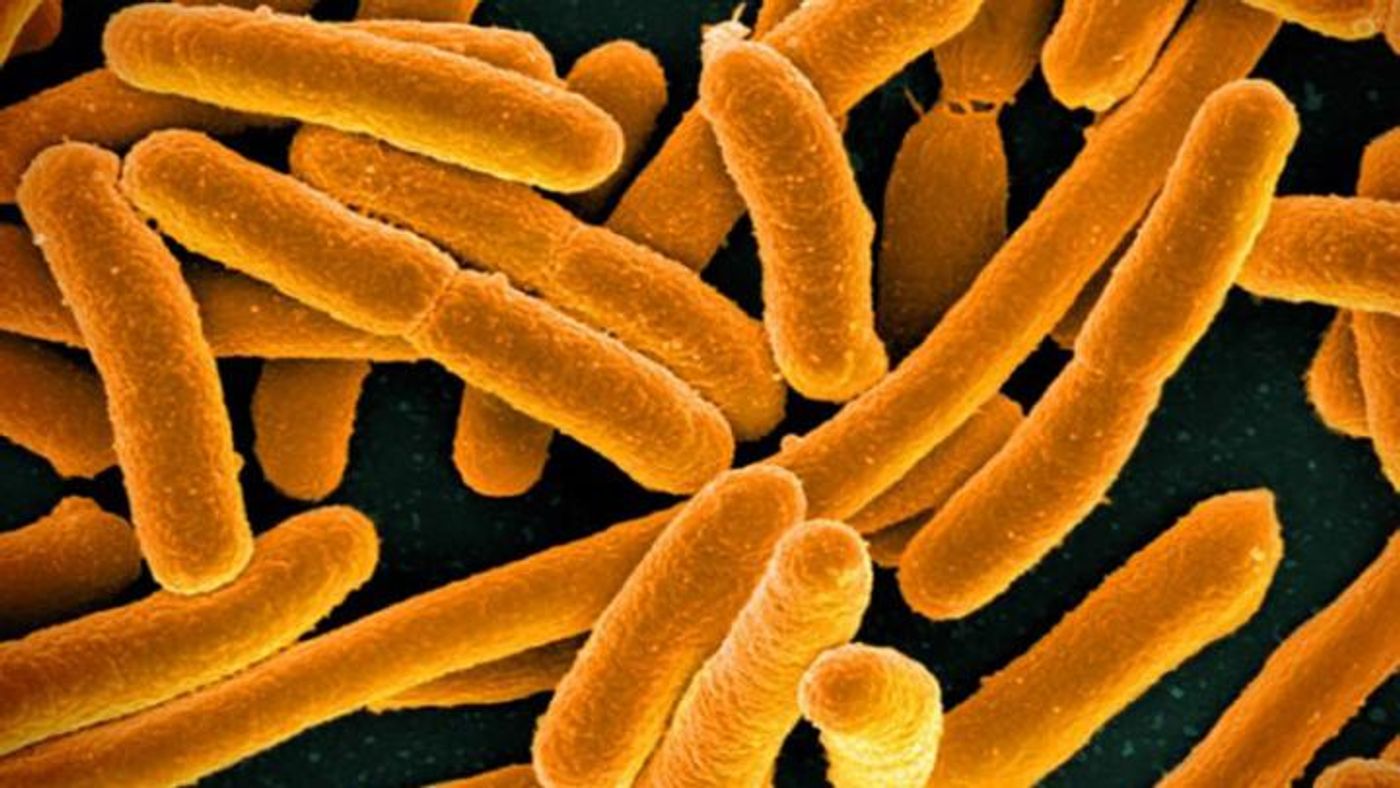
In this work, E. coli bacteria that carried a tetracycline-resistance plasmid and Klebsiella pneumoniae bacteria with a chloramphenicol-resistance plasmid were grown without antibiotics, and after nine days, less than 20 percent of the E. coli and less than 50 percent of the Klebsiella still carried the plasmid. When the bacteria were grown with the antibiotic they resist for 400 generations, they held onto their resistance plasmids even when the antibiotic was removed. After being grown without antibiotics for nine days, over 50 percent of each type of bacteria had retained the plasmid.
"Of course, the cells needed their plasmids to help them survive the antibiotic exposure. But even after we took away that selective pressure, both strains retained their plasmids at significantly higher levels than they had before the antibiotic exposure," said Jordt.
Klebsiella pneumoniae bacteria were shown to easily acquire plasmids from E. coli bacteria when grown together. But if the bacteria had also been exposed to antibiotics, around 1000 times more multi-drug resistant K pneumoniae were created.
"We believe that by stabilizing one plasmid, these mutations make them more likely to stabilize additionally acquired plasmids," said Kerr.
"There are many, many details to be worked out in the future," said Top. "But what we see here is that even short-term exposure to just one antibiotic accelerates the development of multidrug resistance, which should give us pause as we use these drugs in health care, agriculture and other settings."
Sources: AAAS/Eurekalert! via University of Washington, Nature Ecology & Evolution