A research team at Merck (accidentally) uncovered a new antibacterial target - a
riboswitch important for
riboflavin (vitamin B2) biosynthesis. The team screened nearly 60,000 small molecules for antibacterial activity and found one that suppressed bacterial growth by binding directly to the riboswitch.
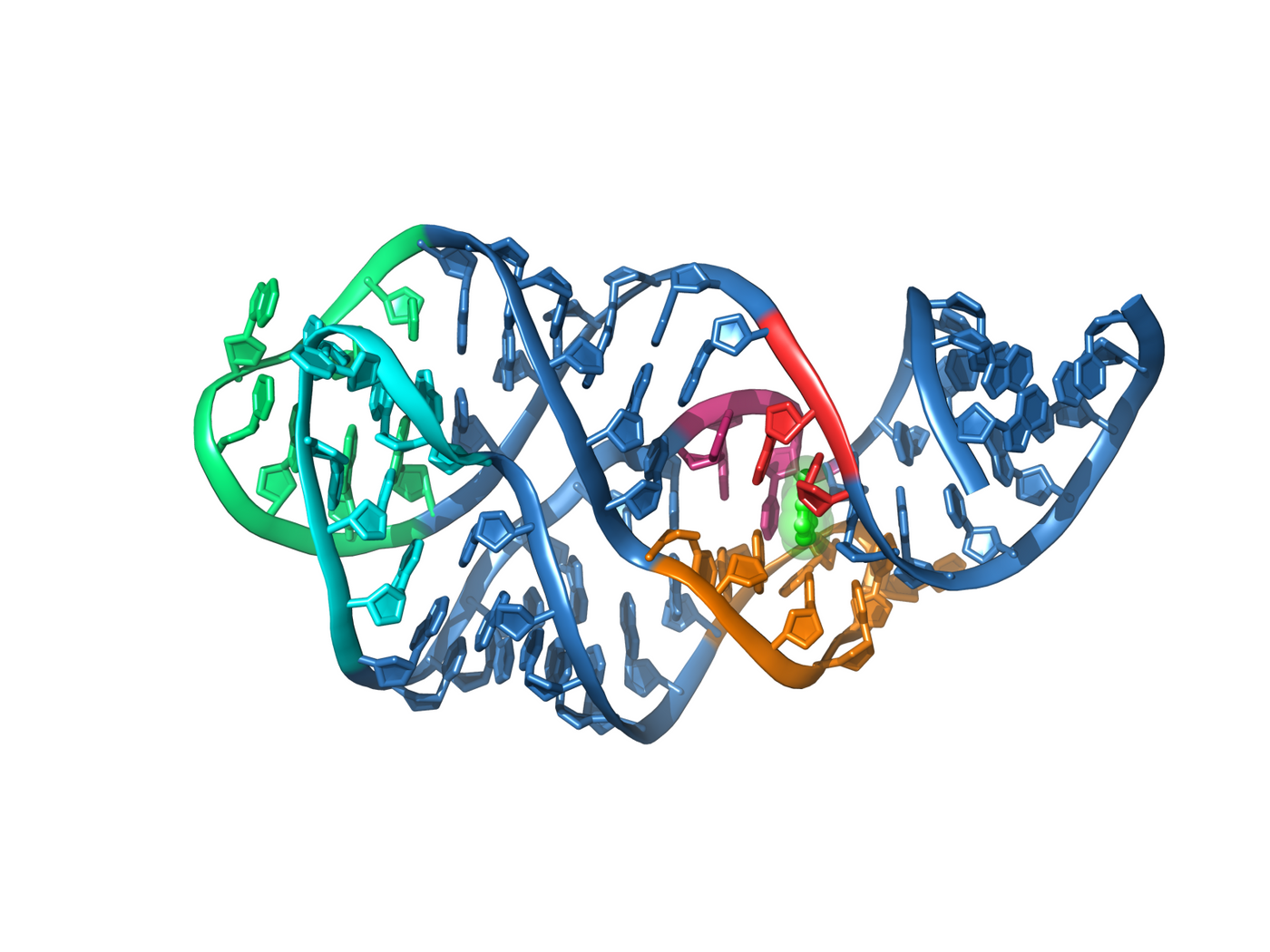
A riboswitch is a segment of messenger RNA (mRNA) that typically interacts with an effector molecule to regulate translation of the mRNA. Riboswitches contain two functional parts, the aptamer, which interacts with the effector molecule, and the expression platform, which directly regulates gene expression. Many riboswitches regulate the production of metabolites such as riboflavin, glutamine, glycine, and lysine. The riboflavin riboswitch binds flavin mononucleotide, which is synthesized from riboflavin, to downregulate the riboflavin synthesis pathway.
The Merck team, led by John Howe, set out to find a drug that would kill bacteria by blocking riboflavin synthesis. This pathway is unique to bacteria, so there would be no off-target effects on humans. To find their drug, they screened nearly 60,000 small molecules for the ability to kill
E. coli in the absence of riboflavin. According to Howe, “if the effect of that antibacterial was suppressed by riboflavin … then we had a good chance that the small molecule … was targeting the riboflavin pathway”.
The group confirmed that ribocil effectively killed
E. coli cells in culture, then tested its activity in a mouse model. After altering its structure to increase its activity, they showed that ribocil decreased the bacterial load in mice.
One (relatively significant) shortcoming is that ribocil was only effective at killing a weakened strain of
E. coli. Wild type cells were able to expel the drug, making them resistant. Despite this drawback, however, the Merck team has shown that riboswitches are viable antibiotic targets.