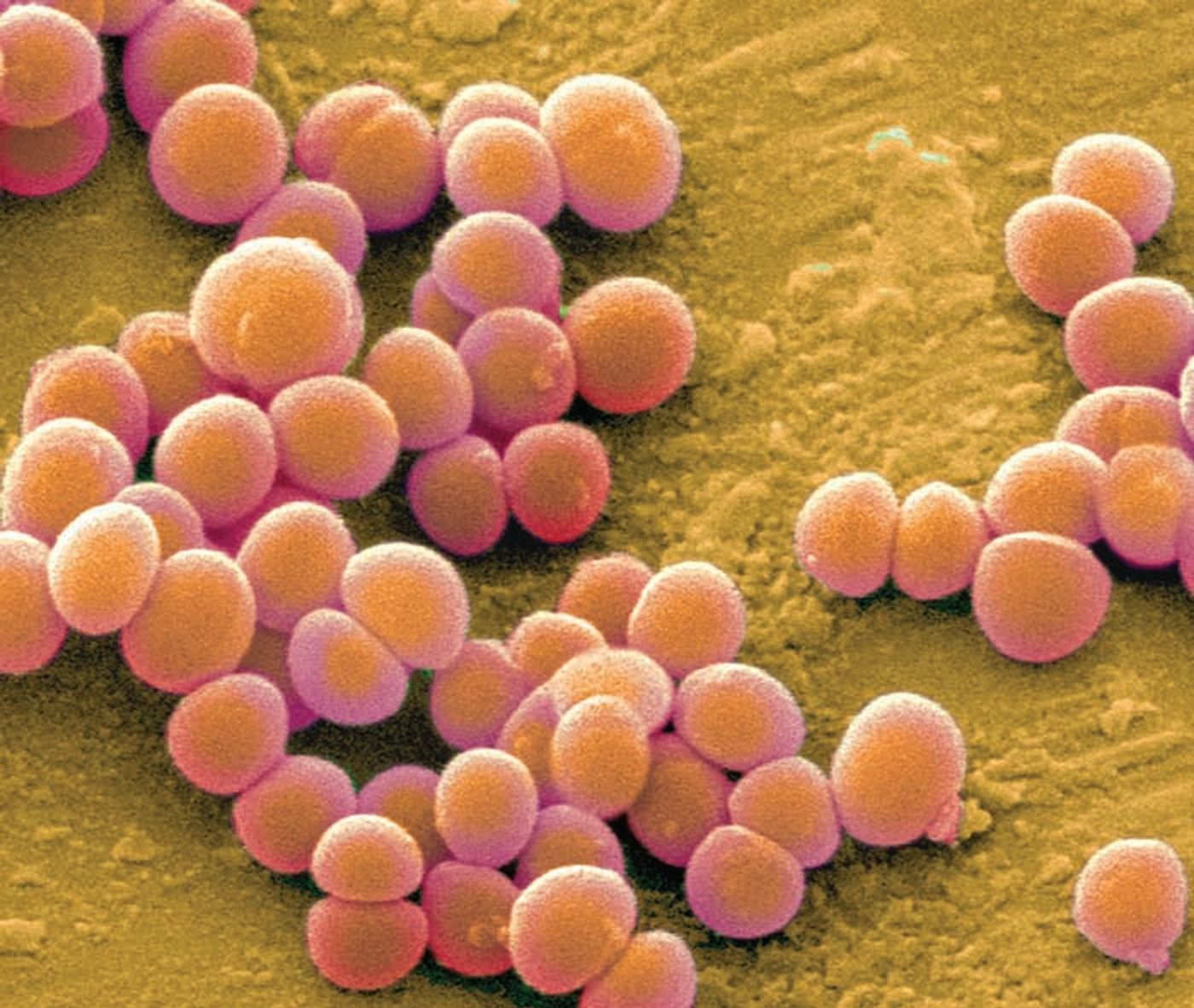
Small genetic changes in Staphylococcus aureus affect how and where these bacteria infect people.
Staph is a normal part of your microbiome - about 29% of the US population carries Staph on their skin or in their noses. (Are you part of the 29%?!) If these cells find their way into your bloodstream, however, you can end up with a life-threatening infection.
Here are some scary Staph statistics. Around 90,000 people are infected with MRSA (methicillin-resistant
S. aureus) in the US each year, and nearly 50% of people report recurring MRSA infections. In England, there was a 548% increase in MRSA-related deaths between 2003 and 2004. Last, but not least, almost 20% of bloodstream infections are caused by
Staph.
According to University of Wurzburg investigator Thomas Rudel, “recent studies have shown that in some cases Staphylococcus strains in the blood of patients differ substantially from those found in the nose … that is astonishing, as both types of bacteria often are very closely related in genetic terms”.
Rudel and colleagues found that one specific mutation, the loss of function of a gene called “repressor of surface proteins” (rsp), allowed Staph to move from the nose into the bloodstream - where it could cause severe infection. What’s strange is that this mutation actually makes Staph less virulent. Essentially, the toxic strains of Staph that live in your nose acquire an rsp mutation, making them less toxic, but better able to enter the bloodstream.
So, how does this all work? The group found that rsp expression helps Staph kill immune cells - by producing toxins in response to hydrogen peroxide (which is produced by neutrophils), for example. When they infected mice with an rsp mutant, the bacteria produced fewer toxins, but were still able cause abscesses. The mutant bacteria were also able to survive in human blood - they weren’t killed by immune cells. This is probably because the rsp mutation allowed the bacteria to survive inside host cells for an extended period of time, but the mechanisms behind this are not clear.
Sources: PNAS,
EurekAlert,
Health Research Funding