Investigators at Baylor College of Medicine have
reported in Cell that they reversed social deficits in mice by adding one specific bacterium to the guts of those rodents. The behavior dysfunction in the animals is a model of autism spectrum disorders (ASDs) in humans.
"Other research groups are trying to use drugs or electrical brain stimulation as a way to reverse some of the behavioral symptoms associated with neurodevelopmental disorders -- but here we have, perhaps, a new approach," explains senior author of the work Mauro Costa-Mattioli, an Associate Professor in the Department of Neuroscience at Baylor College of Medicine.
Epidemiological studies that have determined that obesity during pregnancy may increase the risk of having a baby with neurodevelopmental disorders such as ASDs. Additionally, gastrointestinal disorders and related symptoms are commonly reported in people with ASDs. Research on the microbiome has expanded to many areas, including demonstrating that as diet can alter the gut microbiome, the microbiome can then influence on the brain. So the researchers wanted to investigate whether there might be a connection between the microbiome and ASDs.
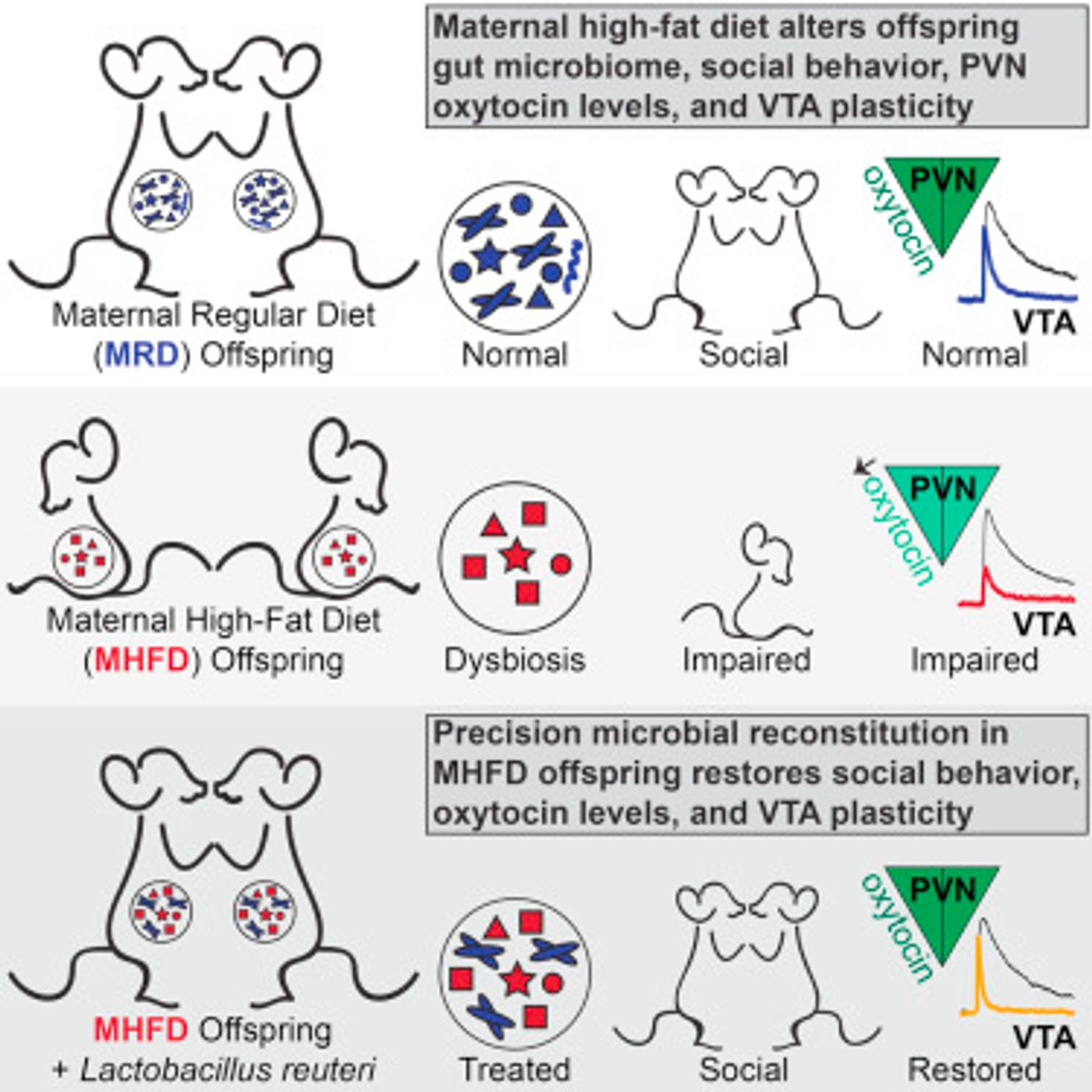
The scientists started by feeding a large group of female mice a high-fat diet, roughly simulating eating multiple fast food meals every day. After breeding these mice, the offspring were kept with their mother for three weeks, and were then put on a normal diet. The researchers observed that these offspring had behavioral deficits; they were not initiating contact or spending much time with their peers. They discuss this research in the video below.
"First we wanted to see if there was a difference in the microbiome between the offspring of mouse mothers fed a normal diet versus those of mothers fed a high-fat diet. “ The team used genetic sequencing to analyze the bacterial composition of the gut. “We found a clear difference in the microbiota of the two maternal diet groups," says the first author of the study, Shelly Buffington, who is a postdoctoral fellow in Costa-Mattioli's lab. "The sequencing data was so consistent that by looking at the microbiome of an individual mouse we could predict whether its behavior would be impaired."
To test whether the microbiome could be causing the behavior issues, they housed mice that were socially impaired and born to dams on a high-fat diet with mice that were born to normal mothers. Mice are coprophagic (they eat each other's feces) and as such, they transfer gut microbiota back and forth. After cohabitation, complete restoration of the gut microbiome was seen, along with a simultaneous improvement in behavior, within four weeks. These observations were confirmed when the team performed fecal-transplants on germ-free mice, establishing causal evidence that an imbalanced microbiome in mice born to dams on a high-fat diet is to blame for their social impairments. It was concluded that one or more bacterial species may be vital for normal social behavior.

The study went further and identified the bacterial species responsible for these changes. Genomic sequencing revealed that Lactobacillus reuteri was more than nine-fold reduced in the microbiomes of mice born to dams fed a high-fat diet.
"We cultured a strain of Lactobacillus reuteri originally isolated from human breast milk and introduced it into the water of the high-fat-diet offspring. We found that treatment with this single bacterial strain was able to rescue their social behavior," Buffington explains. It did not correct the anxiety seen in the mice, however.
Digging deeper, the investigators determined that L. reuteri promotes the production of oxytocin, a hormone that is known to play a critical role in social behavior and has also been connected with autism in humans.
The team thought the reward circuitry in the brains of socially impaired mice was dysfunctional. "We found that in response to social interaction there was a lack of synaptic potentiation in a key reward area of the brain that could be seen in the normal control mice," says Costa-Mattiol. "When we put the bacteria back in the maternal-high-fat-diet offspring, we could also restore the changes in synaptic function in the reward circuitry."
The researchers suggest that probiotic intervention for the treatment of neurodevelopmental disorders in humans is an avenue to explore. "This is where the science is unexpectedly leading us. We could potentially see this type of approach developing quite quickly not only for the treatment of ASD but also for other neurodevelopmental disorders; anyway, this is my gut feeling," elaborates Costa-Mattioli.
Sources:
Cell,
Cell Press via
Science Daily