It’s common knowledge that bacteria replicate by making exact copies of themselves, right? Well, that’s true, most of the time.
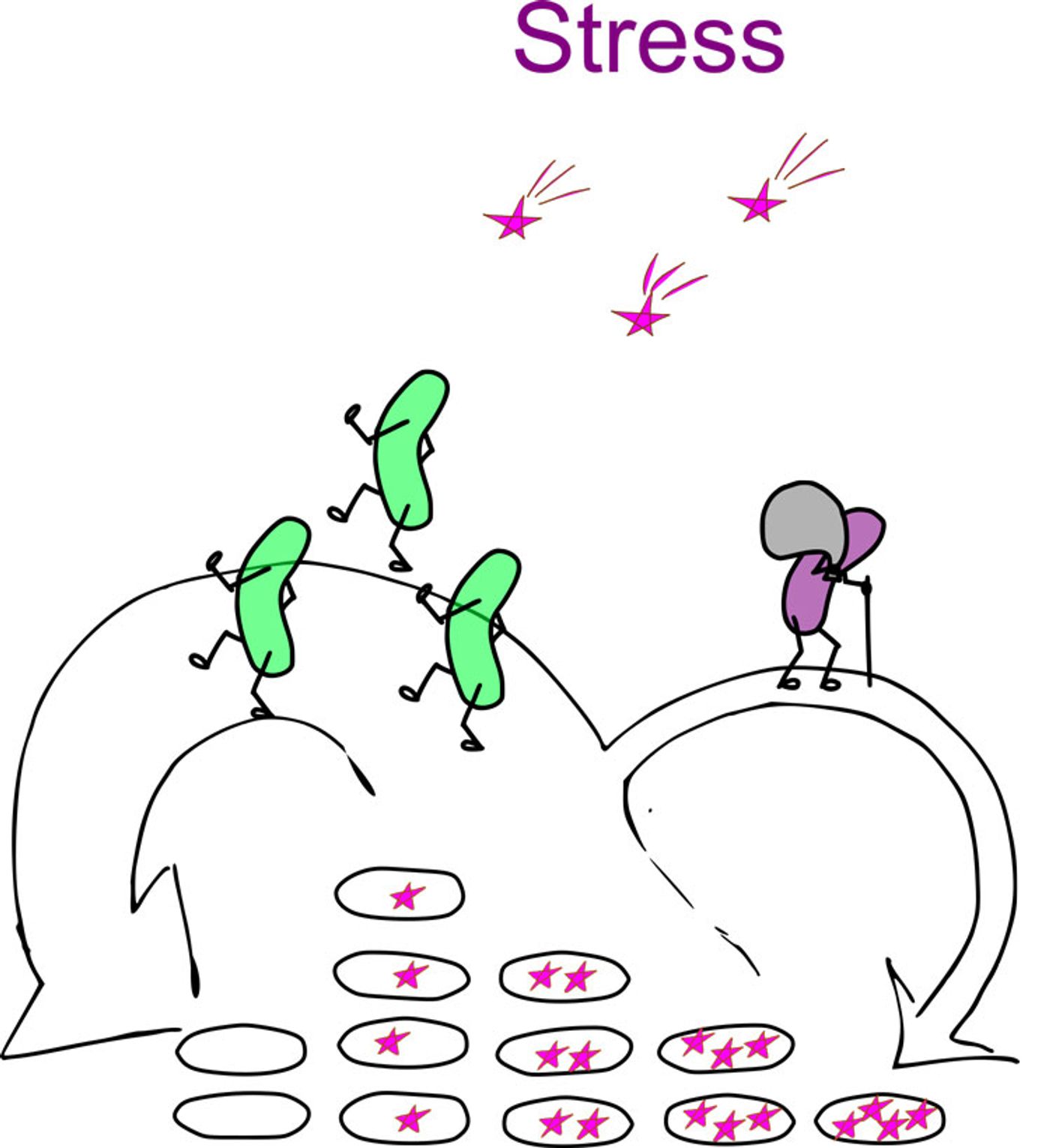
Researchers from the Niels Bohr Institute at the University of Copenhagen, led by Ala Trusina, found that, when stressed, bacteria partition more of their genetic defects into one daughter cell than the other.
Since all organisms accumulate genetic defects as they age (humans included), Trusina reasoned that a colony of bacteria must have some way to minimize passing on these defects in order to keep the population healthy. “We wanted to investigate whether the bacteria divided symmetrically with an equal number of defects in both new individuals or whether they divided asymmetrically with more defects in one new bacteria than the other”, says Trusina.
In other words, are genetic defects spread evenly throughout the population, or are they effectively partitioned into a subset of the population?
What they found was that, when E. coli bacteria were not stressed (they had plenty of food, the temperature was perfect, etc.), mutations were divided symmetrically between daughter cells. That is, genetic defects that accumulated over time were spread equally throughout the population.
On the other hand, if the researchers stressed the bacteria by increasing the temperature or adding antibiotics, mutations were passed on asymmetrically to the daughter cells. The result was that only a subpopulation of bacteria “aged”; these cells also grew more slowly than their “younger” relatives. This way, the colony contained a subpopulation of healthy, super bacteria that could cope with environmental stresses.
Other data from Trusina and colleagues indicate that this aging process is controlled primarily by the environment. “What we have found is that the asymmetry of cell division is not controlled genetically. It is a process that is controlled by the physical environment. Through collective behaviour, the bacterial colony that is exposed to stress can stay young, produce more offspring, and keep the colony healthier. This is completely new knowledge that has never been observed before”, says Trusina.
This type of genetic segregation falls under the umbrella of “asymmetric damage segregation” (ADS). Trusina describes it as “a mechanism for increasing population fitness through non-random, asymmetric partitioning of damaged macromolecules at cell division”. This phenomenon was already reported for protein aggregates in
E. coli - a protein called IbpA aggregates old, damaged proteins at the cell poles. These aggregates become more prominent as cells age.
In fact, ADS may play an important role in diseases such as Alzheimer’s and Parkinson’s - conditions characterized by the accumulation of protein aggregates.
Source: University of Copenhagen,
Cell Systems,
PNAS