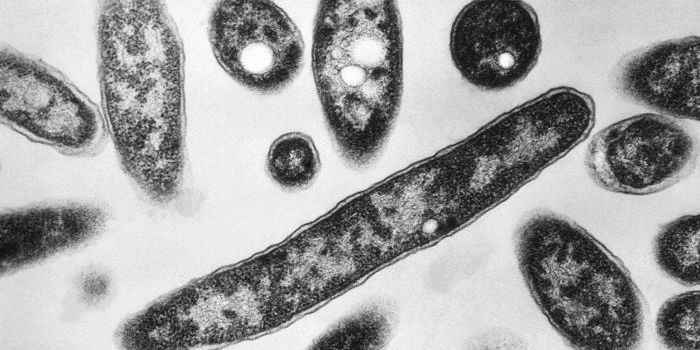
Biofilms form when microbes attach to a surface and adhere to each other, often inside of a matrix created by the cells of the microorganisms. These slimy biofilms are interesting to researchers for many reasons, one of which is because they form inside of the human body, where they can have a deleterious influence. Bacteria that reside in these biofilms are a thousand times less affected by antibiotics, and can thus make infections far more dangerous and tough to treat.
New work performed by scientists at Princeton University and
published in the Proceedings of the National Academy of Sciences has followed a single bacterial cell as it expanded into a mature biolfilm consisting of over 10,000 cells, complete with an ordered structure. These observations could help researchers learn more about how to fight biofilms with drugs, and how the bacteria inside of them behave.
"No one's ever peered inside a living biofilm and watched it develop cell by cell," explained the senior author of the report, Bonnie Bassler, the Squibb Professor in Molecular Biology at Princeton and a Howard Hughes Medical Institute Investigator. "With this paper, we can now understand for the first time how communities of bacteria form a biofilm."
A former Princeton post-doc, Knut Drescher, developed a cutting edge microscopy method that enabled single cells to be imaged, thus allowing the researchers to watch the development of the biofilm as it happened.
"We have used a state-of-the-art technique to see into the core of a living, growing biofilm," said the lead author of the new study, postdoctoral research associate Jing Yan.
"The research that produced this paper sits at the frontier between materials science, engineering, physics and biology and represents a fantastic collaboration across Princeton University," said Bassler.
For this research, the investigators utilized Vibrio cholerae as a model; the pathogen has been studied in depth and is relevant as a threat to human health; it causes cholera, a diarrheal disease. Vibrio is a rod-shaped, curved bacterium that resides in brackish water or saltwater as a free-swimming cell. When it attaches to a surface that can provide it with sustenance, such as a crab shell or shrimp, or in a human intestinal cell, the microbe attaches and starts to reproduce. The population of cells secretes a sticky substance that acts like glue, keeping the film attached and protecting the bacteria from competitors.
Research in this area has been impeded by a lack of optical resolution; the behavior of individual cells was obscured by the mass, and the behavior of individual cells could not be differentiated from that of neighbors. To address that issue, the scientists took several approaches. They genetically altered their strain of bacteria to produce glowing proteins after fluorescent excitation. The selected proteins were picked because they would emit enough light to allow individual cells to be discerned, while keeping light that could pose a danger to the cells to a minimum.
The team also took advantage of confocal microscopy for their work to hone in on small parts of their specimen. After making hundreds of observations, they were able to create a three-dimensional reconstruction of the whole specimen. "It's like looking deep into the interior of a biofilm without having to slice it open," explained Yan. In the video from their work above, the V.cholerae biolfilm is seen.
Computer algorithms aided their work even more, enabling the team to see differences in bunched up sources of light, like those in the dense biofilm of V. cholerae.
The investigators saw an incredible phenomenon. The bacteria began by horizontally expanding on the provided surface, and then as each cell split, daughter cells anchored onto the surface next to their parents. As the offspring increased, the cells at the source of the growing colony were continuously squeezed until they had to detach from the surface. At that point, the two-dimensional film became a three-dimensional mess, all stuck together by the matrix of the biofilm.
The researchers investigated the genetic events underlying this behavior as well. They found that one gene, RbmA, is critical to the connection of cells that results in biofilm. After deactivating that gene, the biofilm made by the altered cells was diffuse and floppy. Correcting the level of RbmA restored biofilm to its normal structure. The scientists think RbmA could be a target for interventions that interfere with biofilm formation. The mutant cluster growth is shown in the video below.
The investigators are working to measure the physical forces that cells experience as they lift from the center in an attempt to reveal the overall mechanics. "We are currently trying to develop a mathematical model for how the bacterial colony grows in time and how the spatial features are linked to typical mechanical features of the biofilm," explained Stone.
The researchers also want to apply their techniques to the study of other bacteria like Pseudomonas aeruginosa, the primary cause of deadly lung infections in cystic fibrosis, or Staphylococcus aureus, commonly known as staph. It is expected that these biofilms will exhibit different propoerties.
"This paper opens up a world to us that was never before accessible: the inside of biofilms. My hope is that other researchers working on biofilms will want to use this technology to rapidly move the field forward," concluded Bassler.
If you’d like to know more about biofilms, check out the video above from the National Science Foundation.
Sources:
AAAS/Eurekalert! via
Princeton University,
PNAS