Bone Marrow Drug Dramatically Reverses Baldness
A new drug may lift hopes for many people plagued with a certain type of hair loss. In clinical trials, researchers have demonstrated dramatic reversal of hair loss in 75 percent of patients after treatment with ruxolitinib, a JAK inhibitor. While promising, there are several caveats to the success of this drug. However, researchers and hair loss patients are hopeful the new drugs can soon be adapted or translated to other more common types of hair loss.
Our hair is extremely personal and is part of our expression of our identity. But for the millions of people suffering from hair loss (alopecia), this form of expression is limited. One type of alopecia, alopecia areata, results from the autoimmune attack on hair follicles.
Previously, researchers studying this disease reported that certain immune cells in the Janus kinase (JAK) pathway seemed to be responsible for the hair loss. They hypothesized that inhibiting the JAK family of enzymes could reawaken the damaged and dormant hair follicles to induce hair growth.
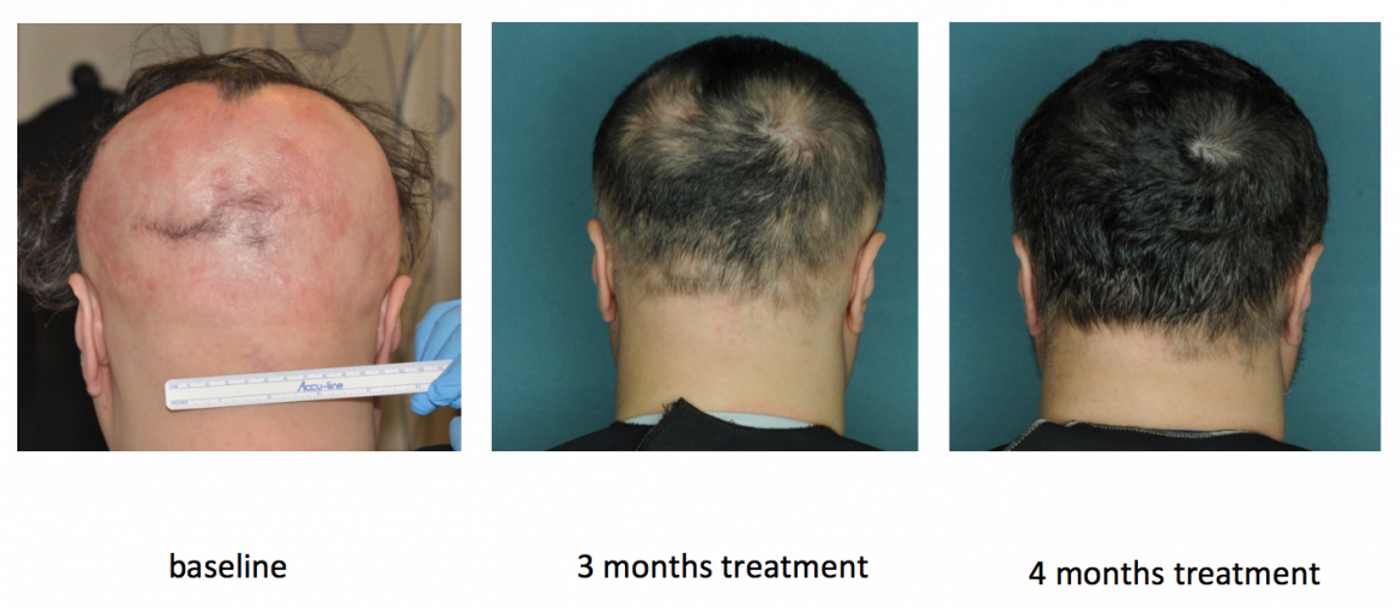
Indeed, in a small clinical trial with 12 patients with mild to moderate alopecia areata, the results seemed to corroborate this hypothesis. The patients were prescribed 20 mg of ruxolitinib, an FDA-approved JAK inhibitor, for 6 months. At the end of the study, 9 out of 12 patients had greater than 50 percent of hair regrowth. In fact, the results were closer to 95 percent of hair regrowth among those who responded to treatment.
“Although our study was small, it provides crucial evidence that JAK inhibitors may constitute the first effective treatment for people with alopecia areata,” said Julian Mackay-Wiggan, the study's lead author. “This is encouraging news for patients who are coping with the physical and emotional effects of this disfiguring autoimmune disease.”
Importantly, hair regrowth was dependent on taking ruxolitinib continually. Among those who stopped medication, hair loss returned. “Our findings suggest that initial treatment induces a high rate of disease remissions in patients with moderate to severe alopecia areata but maintenance therapy may be needed,” said Mackay-Wiggan.
Ruxolitinib is already approved by the FDA for other diseases, such as bone marrow malignancies and rheumatoid arthritis. And while there were no major side effects noted in this small study, researchers found that participants had a weaker immune system while on the drug, which made them susceptible to more illnesses.
As dramatic as the before-and-after evidence may be, the celebration does not yet include people who suffer from androgenic alopecia (also known as male or female pattern baldness), which is the most common kind of baldness. However, researchers agree that this is an excellent step forward in understanding how to treat the other types of hair loss conditions.
“We expect JAK inhibitors to have widespread utility across many forms of hair loss based on their mechanism of action in both the hair follicle and immune cells,” said co-author Dr.Angela M. Christiano, a professor of genetics and development and dermatology at Columbia.
Additional sources: Columbia University Medical Center
Image credit: Mackay-Wiggan et al. 2016, JCI Insight